Home » Phosphorus (Page 2)
Category Archives: Phosphorus
The Sufficiency versus Replacement tipping point.
Being educated in the realm of Soil Fertility at Oklahoma State University by the likes of Dr Gordon Johnson and Dr. Bill Raun, Brays Nutrient Mobility Concept and Mitscherlich’s Percent Sufficiency Concept are ingrained in my psyche. In class the concept of Build and Maintain for phosphorus fertilizer management was just briefly visited and not discussed as a viable option. For anyone in the corn belt, and some Okies, reading this that may seem unusual. But when I was in school on average in Oklahoma there was about 100-200 K acres of 100 120 bpa (bushel per acre) corn, 300-400 K acres of 40-50 bpa sorghum, and over 5 million acres of 20-30 bpa wheat. In a state with those average yields, replacing P removed by the crop was not a major concern.
But times are changing. There is more corn and soybean planted and the achievable yields of all crop are increasing. While the average winter wheat producer should not be worried about replacement rates of P there is a growing group of producers that should. This blog will discuss the scenarios in which sufficiency rates are best and those in which replacement should be considered. The OSU factsheet PSS-2266 goes in-depth on each of these methods.
Applying P based on sufficiency will increase soil test P levels in a low yielding environment. For example on a 20 bpa wheat field that starts out with a soil test P level of 0. Using the sufficiency recommendation each year the soil test value will reach 20 ppm (40 STP) in 20 years. A 30 bpa field would take 30 years. Yes that is a long time but the soil test value is increasing a little each year. The point of 20 ppm is important because at that level the crop is 95% sufficient, meaning if no P is added the crop will only reach 95% of the fields yield potential.
Using a mass balance approach we can determine at what point does the crop remove more than we can supply with in or near furrow starter fertilizer. Table 1 shows the values I am using for the discussion. The first column is just the average amount of P removed per bushel of grain, most of our grains fall in the .4 to .5 lbs P per bushel range. The second column is the soil test value at which P level is said to be at 90% sufficient. The reason this column is included is that the P2O5 reccomendation for this P level fits into the starter rate for all crops. The low high starter rates are the typical range of P2O5 that is delivered within the safe range (N based) and what I see as the common rates. These values may be above or below what you use.

Table 1. Values used to create Table 2. Phosphorus per bushel of grain. Mehlich 3 soil test value (ppm) at which crop is determined to be 90% sufficient, typical range of P2O5 applied with starter fertilizers, recommended P2O5 rate when soil test P is at 90% sufficiency.
Table 2 is pretty simple but it is the center point of this article. The one caveat I need to add is this assumes strip till or 2*2 / 3*2 is not being used. Table 2 is using the starter range and removal value to determine the yield level the starter can support. The first take on this table may provide some hint on why in a state with 5 million acres of wheat averaging 36 BPA the state soil fertility specialist didn’t focus on replacement rates. In fact for most for most the the wheat ground P application is higher than removal and P levels are slowly increasing. The big take home from this table should be is my yield level outside this window? If so do not immediately go out in crease your P rates but do take a close look at your system as a whole. Take a close look at your cropping system, not just one seasons but look at a three or four year cycle. Add up P applied and P removed, are you positive or negative net balance? If you are negative take a long hard look at your soil test over time. Some soils can supply a large amount of P even if you are removing more than you apply. Other soils will be rapidly drawn down. Regualr soil testing allows for producers to keep an eye on these values.

Table 2. Yield level (bushels per acre) at which P removal is equal to P added in starter fertilizer application.
In the end even if the production warrants the use of replacement rates, the current market may not. For more on that read https://osunpk.com/2016/08/27/now-may-not-be-the-time-for-replacement/.
Speaking of market currently both soybeans and cotton are getting a lot of attention due to how the economics is penciling out. Soybean is a “heavy” P crop pulls .8 lbs per bpa while cotton removes 13 lbs per bales. Both of these crops are salt sensitive and the rate of inforrow is typically quite low providing only about 6 lbs when on 30″ rows. If you are growing beans or cotton make sure you account for their removal when you talley up your system.
Below is a table that I wanted to add, well because I like it. This table illustrates that buildup, and drawdown, rate is heavily impacted by existing soil test value. In short it takes a lot more fertilizer P to raise soil test p levels in a very low P testing field than it does when soil test P is closer to optimum, 19 lbs per 1 lb at STP of 10 and 5 lbs per lb when STP is 65. The exact rate changes by soil type and the same holds true to drawn down via crop removal.

Amount of P2O5 above crop removal needed to increase soil test phosphorous based upon initial soil test results.
Adapted From http://www.spectrumanalytic.com/support/library/ff/Soil_test_P_and_K_buildup_and_drawdown.htm “Drawdown of Soil Test Phosphorus and Potassium Levels by Alfalfa, K.L. Wells & J.E. Dollarhide, Univ. of Kentucky, Soil Science News & Views, Vol. 21, No. 3, 2000”
Any questions or comments? Feel free to contact me at b.arnall@okstate.edu
Precision Nutrient Management in Forage Systems
Published in Progressive Forage http://www.progressiveforage.com/ 9.1.2016
First, let’s agree the term “precision” is relative. Forage is a diverse system with an even more diverse set of management strategies. Regardless, every manager should be constantly striving to improve the precision in which nutrients are managed. The ultimate goal of any precision nutrient management tool should be this: producing the highest quality output (in this case forage) with the least amount of input – ultimately, optimizing efficiencies and maximizing profits. Within this readership there are those who are soil sampling at a 1-acre resolution and others who have likely not pulled a soil sample in the past decade. For both spectrums we can make improvements – let’s start basic and move forward.
A soil sample should the basis for all nutrient management decisions. Is soil testing a perfected science? No, far from it. However, there must be a starting point. A soil sample is that first bit of information we can start with and the basic data collection for precision ag to make improved management decisions. When fertilizer is applied without a recent soil sample, it is done based upon pure guesswork. How many other management decisions are made on a farm or ranch by a guess?
The composite soil sample is a great start, but it is just that – a start. While there are some soils that are very uniform most are extremely variable. In a survey of 178 fields in the southern Great Plains on average the soil pH was 6.12; phosphorus (Mehlich 3 phosphorus [M3P] and Bray 1 phosphorus [B1P]) was 28 ppm while soil test potassium averaged 196 ppm. So on the average the primary components of soil fertility were okay. However, on average the 178 fields had a range in soil pH of 1.8 units, M3P and B1P both had range of a 52 ppm and STK had a range of 180 ppm.
Table 1 shows the minimum and maximum soil test values for the 178 fields.
| Average | Range | Min | Max | |
| Soil pH | 6.12 | 1.77 | 5.23 | 7.01 |
| Phosphorus | 28 | 52 | 2 | 54 |
| Potassium | 197 | 180 | 107 | 287 |
| Sulfur | 15 | 24 | 3 | 27 |
| Organic matter | 1.9 | 1.2 | 1.3 | 2.5 |
This data helps support the concept that we should find ways to increase the resolution or decrease the number of acres represented by a single soil sample. Increasing soil sample resolution is typically done using one or two methods – zone or by grid.
Zone sampling
The basis of a zone sample is creating a smaller field. The biggest question with zones is how to draw the lines. There are dozens, if not hundreds, of possible methods, each having their own reasons and benefits. My basic recommendation is that before lines are drawn goals have to be established. For example, if phosphorus or soil pH management is important, the basis for the lines should be soil based. This could be based on soils map, soil texture, slope and on and on. If the target is improved nitrogen management, then the reason for drawing lines should be yield based. This could be based on yield maps, aerial images, historic knowledge or many soil parameters.
Why does it matter? Two reasons: First, across the broad spectrum of soils and environments two nutrients are hardly spatially correlated, which means the zone that is best at describing phosphorus variability does an extremely poor job describing potassium variability. Second, more theoretically the demand for nutrients are driven by different factors. Phosphorus (a soil immobile nutrient) fertilizer need is driven by the soil P concentration (look up Brays Sufficiency Concept). Many use yield as a parameter for phosphorus application, but this is not a plant need or even a yield maximizing practice. Fertilizing based on removal is done to prevent nutrient mining. However, nitrogen (a nutrient mobile in the soil) fertilizer need is based on yield and crop removal. Hence, the common Land Grant University N and sulfur recommendations are yield goal based.
Grid sampling
To be honest even the experts disagree on the hows, whys and ifs of grid sampling. I like data, therefore I naturally lean towards grid sampling if the field warrants it. For me, the biggest benefit of grid over zone sampling is that soils data from zone samples are biased to whatever parameter was set for the zone and therefore any resulting map for all nutrients must reflect the original zones. In a grid, each data point is independent therefore the maps of each nutrient can be independent, and (the science tells us) in most cases nutrients are independent of each other.
Ideally two pieces of information are available for determining whether a field is grid sampled or not. The first piece of information is a yield map from any previous crop. If yield is fairly uniform, I question the need for variable rate management, much less the expense of grid sampling. Regardless of the sampling method zone or grid, the discussion is moot if spatial variability does not exist across the field. However, many forage producers may not have access to this kind of data.
One of the most useful decision aid tools for grid sampling is the composite soil sample. The reason is simple statistics: A composite sample should be a representative average of the field. If the data is normally distributed, that means half of the field is above and half the field is below the sample average. So the optimum fields to grid are those in which an input falls at the point in which the benefit of applying is in question, because it suggests that approximately half the field needs the inputs while the other half likely does not. It is in this scenario that the return on investment can be greatest. As with pH, for example, fields with a very low value should have a flat broadcast application and should be sampled again at a later date. Fields with a composite pH well above 6.0 will unlikely have enough acres needing lime to warrant sending out an applicator.
Is grid sampling a lifelong activity? No. The initial activity of grid sampling will provide both an indicator of the variability level and overall needs of the field. From that point, decisions can be made and actions taken. Identify the greatest limiting factor in the field based on the samples, and focus on impacting change upon it. Zone sampling in subsequent years can be utilized to document change. When that issue is resolved, move to the next factor. It may require grid sampling again or using the original grid to develop new management zones. For instance, if the greatest issue first identified on the field is soil acidity then after the soil pH is neutralized the field should be grid sampled again. The reason is for this is that changing soil pH will influence many nutrients and the amount of change is not consistent but dependent upon many other factors.
In precision ag we tend to look at layers, yield, soil, etc. However, none of these tell the whole story independently. An area in a field may have moderate soil fertility and be under producing. Using the data collected the decision may be made to increase inputs; yet, the issue is a shallow restrictive layer limiting production. Therefore, the extra inputs will be of no benefit and could even further reduce production. It is at this point I like to bring out the importance of “getting dirty.” There is no technology that can take the place of “boots on the ground” agronomy.
For producers who have historically preformed intensive soil sampling there is still room for improvement. Soil testing and nutrient management is not an exact science; in fact, it was originally built for broad sweeping, statewide recommendations. As technology advances and inputs can be applied at sub-acre resolutions, all of the environment (weather, soil) by genotype inactions becomes more evident.
The next step in precision ag is to develop recommendations by upon site specific crop responses. This is where nutrient response strips can further improve nutrient use efficiencies and crop production. In Oklahoma, nitrogen-rich strips are applied across fields (grain and forage) to determine in-season nitrogen needs. Having a strip in the field with 50 to 100 extra units of N acts as a management tool which takes into account soil, environment and plant need. If the strip is visible the field or zone needs more N, if it is not visible then the crop is not deficient and at that point in the season does not need more N. Producers have taken this approach for N and adopted it for P and K with strips across the field with a zero and high rate of either nutrient. After a few seasons, responsive and non-responsive zones are developed and P and K applications are managed accordingly.
One misconception of precision ag is that the end result should be a field with uniform yield from one corner to the other. This is often not the case; in fact, in many cases the variability in production across the field can be increased. Theoretically, precision ag is applying inputs at the right rate in the right place. This means areas of the field which are yield limited due to underlying factors which cannot be managed have a reduction in inputs with no effect on yield. Other areas of the field have not been managed for maximum production therefore an increase inputs result in increasing yield widening the gap between the low and high yield levels.
Regardless of where a producer currently sets on the technology curve, there are potential ways to increase productivity and efficiency. There is nothing wrong with taking baby steps; it is often the simple things that lead to the greatest return.
Now may not be the time for Replacement
For phosphorus (P) and potassium (K) fertilizer management there are three primary schools of thought when it comes to rate recommendations. The three approaches are Build-up, Maintenance/Replacement, and Sufficiency. There is a time and place for each one of the methods however the current markets are making the decision for the 2016-16 winter wheat crop a very easy one. The OSU factsheet PSS-2266 goes in-depth on each of these methods. For the rest of the blog I will use P in the conversation but in many scenarios K should/could be treated the same.
Build-up is when soil test is below a significant amount of fertilizer, about 7.5 lbs P2O5 per 1 ppm increase, is added so that soil test values increase. This method is only suggested when grain price is high and fertilizer is relatively cheap. Given the market, this is a no go. The two most commonly used methods of recommendation are Replacement and Sufficiency. In the replacement approach if the soil is at or below optimum P2O5 rate it based upon replacing what the crop will remove. The sufficiency approach uses response curves to determine the rate of P that will maximize yield. These two values are typically quite different. A good way you boil the two down is that replacement feeds the soil and sufficiency feeds the plant.
Oklahoma State Universities Soil, Water, and Forage Analytical Lab (SWFAL) provides recommendations utilizing sufficiency only while many private labs and consultants use replacement or a blended approach. Some of this is due to region. Throughout the corn belt many lease agreement contain clauses that the soil test values should not decrease otherwise the renter pays for replacement after the lease is over. For the corn belt both corn and soybean can be expected to remove 80 to 100 pounds of P per year. Conversely the Oklahoma state average wheat crop removes 17 lbs P a year. In areas where wheat yields are below 40 bushel per acre (bpa) using the sufficiency approach for P recs can increase soil test P over time.

This conceptual soil test response curve is divided into categories that correspond with below opti-mum, optimum and above optimum soil test values. The critical level is the soil test level, below which a crop response to a nutrient application may be expected, and above which no crop response is expected. At very high soil test levels crop yield may decrease.
*Rutgers Cooperative Extension Service FS719
Back to subject of this blog, consultants, agronomist, and producers need to take a good look at the way P recs are being made this year. Profitability and staying in the black is the number 1, 2, and 3 topic being discussed right now. The simple fact is there is no economic benefit to apply rate above crop need, regardless of yield level. The figures above demonstrate both the yield response to fertilizer based upon soil test. At the point of Critical level crop response / increase in yield is zero. What should also be understood is that in the replacement approach P fertilizer is still added even when soil test is in Optimum level. This also referred to as maintenance, or maintaining the current level of fertility by replacing removal. If your program is a replacement program this is not a recommendation to drop it completely. Over a period of time of high removal soil test P levels can and will be drawn down. But one year or even two years of fertilizing 100 bpa wheat based on sufficiency will not drop soil test levels. On average soils contain between 400 and 6000 pounds of total phosphorus which in the soil in three over arching forms plant available, labile, and fixed. Plant available is well plant available and fixed is non plant available. The labile form is intermediate form of P. When P is labile it can be easily converted to plant available or fixed. When a plant takes up P the system will convert labile P into available P. When we apply P fertilizer the greatest majority of was is applied makes it to the labile and fixed forms in a relatively short period of time. For more in-depth information on P in the soil you can visit the SOIL 4234 Soil Fertility course and watch recorded lectures Fall 2015 10 26-30 Link .
How to tell if your P recs have a replacement factor, not including calling your agronomist. First replacement recs are based on yield goal, so if you change your yield goal your rate will change. The other and easier way is to compare your rates to the table below. Most of the regional Land Grant Universities have very similar sufficiency recs for wheat. Another aspect of the sufficiency approach is the percent sufficiency value itself. The sufficiency can provide one more layer in the decision making process for those who are near the critical or 100% level. Response and likelihood of response to P is not equal. At the lowest levels the likelihood of response is very high and the yield increase per unit of fertilizer is the greatest. As soil test values near critical (32.5 ppm or 65 STP) the likelihood of response and amount of yield increase due to fertilizer P decreases significantly. At a STP of 10 the crop will only produce 70% of its environmental potential if P is not added while at a STP of 40 the crop will make 90% of its potential. The combination of % sufficiency and yield goal can be used to determine economic value of added P.

*From Oklahoma State University Soil Test Interpretations. Fact Sheet PSS-2225
*Based on Mehlich 3
*PPM (parts per million) is used by most labs
*STP (soil test P) is a conversion used by some Universities. Equivalent to pounds per acre.
* for a 0-6” in soil sample PPM * 2 = STP.
This data is available from OSU in multiple forms from the Factsheet PSS-2225, the SWFAL website, Pete Sheets quick cards, and the Field Guide App.
This year with margins tight soil testing is more important than ever before. Knowing the likelihood of response and appropriate amount of fertilizer to apply will be critical maximizing the return on fertilizer invest while maximizing the quality and amount of grain we can produce. Visit with your consultant or agronomist to discuss what the best approach is for your operation. Lets ride this market out, get the most out of every input and come out of this down cycle strong.
Feel free to contact me with any questions you may have.
Brian
b.arnall@okstate.edu
DAP vs MAP, Source may matter!
Historically the two primary sources of phosphorus have had different homes in Oklahoma. In general terms MAP (11-52-0) sales was focused in Panhandle and south west, while DAP (18-46-0) dominated the central plains. Now I see the availability of MAP is increasing in central Oklahoma. For many this is great, with MAP more P can be applied with less material. which can over all reduce the cost per acre. There is a significant amount of good research that documents that source of phosphorus seldom matters. However this said, there is a fairly large subset of the area that needs to watch what they buy and where they apply it.
If you are operating under optimum soil conditions the research shows time and time again source does not matter especially for a starter. In a recent study just completed by OSU multiple sources (dry, liquid, ortho, poly ect ect) of P were evaluated. Regardless of source there was no significant difference in yield. With the exception of the low pH site. The reason DAP was so predominate in central Ok, soil acidity. See an older blog on Banding P in acidic soils.

Figure 1. The cover of an extension brochure distributed in Oklahoma during the 1980s.
When DAP is applied, the soil solution pH surrounding the granule will be alkaline with a pH of 7.8-8.2. This is a two fold win on soil acidity aka aluminum (Al) toxicity. The increase in pH around the prill reduces Al content and extends the life of P, and as the pH comes back down the P ties up Al and allows the plant to keep going. However, the initial pH around the MAP granule ranges from an acid pH of 3.5-4.2. There is short term pH change in the opposite direction of DAP, however the the Al right around the prill becomes more available and in theory ties up P even faster.
Below is a table showing the yield, relative to untreated check, of in-furrow DAP and MAP treatments in winter wheat. The N401 location had a ph 6.1 while Perk (green) has a pH of 4.8. At Perkins in the low pH, both forms of P significantly increased yeild, almost 20 bushel on the average. DAP however was 5 bushel per acre better than MAP. At the N40 site the yield difference between the two sources was 1 bushel.
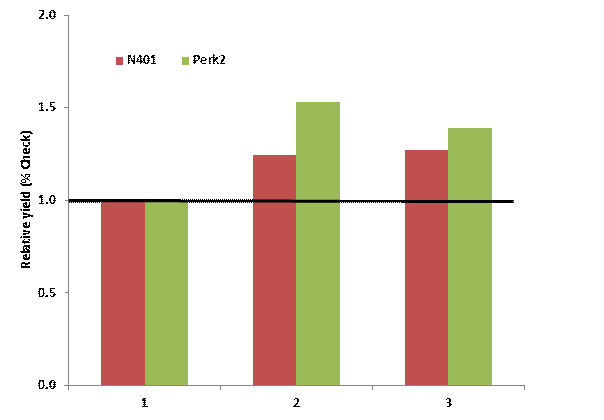
Relative yield winter wheat grain yield MAP and DAP both applied at equal rates of P (32 lbs P2O5 ac) when compared to a untreated check.
In general it can be said that in acid soils DAP will out preform MAP while in calcareous high pH soils MAP can out preform DAP. So regarding the earlier statement about the traditional sales area of MAP or DAP if you look at the soil pH of samples went into the Oklahoma State University Soil, Water, and Forage Analytical lab the distribution makes since.

Average soil pH of samples sent into OSU soil water forage analytical lab by county.
In the end game price point and accessibility drives the system. In soils with adequate soil pH levels, from about 5.7 to around 7.0, get the source which is cheapest per lbs of nutrient delivered and easiest to work with. But if you are banding phosphorus in row with your wheat crop because you have soil acidity, DAP should be your primary source.
Canola fertilizer rates when skip row seeding.
When drilling canola a common strategy to improve seeding rate accuracy is to only use every other row which effectively doubles the rate of seed going through each meter. There are also many producers who utilize air seeders and just prefer the wider spacing. Every season I get several questions about determining total fertilizer rates if the seed is dropped every other row but fertilizer is dropped every row. Regardless of whether or not fertilizer goes down every row it is important that the amount of salts placed with seed does not exceed the limit. The table below provide the limits in terms of lbs of salt per acre. If using 18-46-0 (DAP) or 11-52-0 (MAP) this is equivalent to pounds of N per acre. However if the fertilizer you use contains potassium (K) or sulfur (S), those have to be considered. An easy rule of thumb for determining total salt level of a fertilizer is pounds of N + K + 1/2 S.

Maximum amount of salt that can be applied in furrow with canola seed. Application rate should be at or below this value.
In a scenario in which canola is seeded in skip rows but every row will get fertilizer the total amount of fertilizer can be doubled. For example on a 15″ row spacing the max salt rate is 5 lbs per acre. If you were using DAP as your starter that maximum rate to place in furrow would be 28 lbs of DAP per acre. If using a drill set of 7.5″ spacing and putting fertilizer down every row the max rate would increase up to 56 lbs DAP per acre.

Seed, colored blue, is placed in every other row while an equal amount of fertilizer, dark grey circles, is placed in every row.
Some producers may have the capability of applying different rate in every other row. In this scenario it is important to maintain that safe rate in the seed furrow. In the opposite row, fertilizer rate can go as high as you wish or the equipment can handle.

Seed, colored blue, is placed in every other row while fertilizer, dark grey circles, is placed in every row. In this scenario a high rate is placed in the row without seed.
Now the big question is, “Is between row fertilization a good idea?” While we do not have results on this style of application (trials will be going out this year) we can draw upon upon similar work in other crops. For me the best win would be the second scenario in which a higher rate could be place between the rows. In this row I would use a urea and DAP blend. Any time we can put urea below the soil surface its a win and in fields with very soil soil test phosphorus (P) it would create something similar to the deep P bands once popular in corn production. Now if the field had adequate soil test P, I would focus on urea between rows. Keep in mind it is never a good to place urea in furrow with canola seed. For the average producer who is using a box drill the first scenario is the only option. In this case the rate of the between row bands will be reduced however I still believe on fields with very low soil test P this is potentially a great way to get the rest of it on. Remember if on 15″ and using DAP max rate only gets 12.9 lbs of P2O5 down. If fertilizer is dropped down every tube that number increases to about 26 lbs P2O5, which is still not enough for fields with low soil test P, but is better. With hope we will have some good results to share from the 2015-2016 canola crop.
Nutrient Products: Stabilizers, Enhancers, Safeners, Biologicals and so on.
In this blog I am not going to tell you what to use or what not to use. In fact I will not mention a single product name. What I will do is hopefully provide some food for thought, new knowledge and direction.
First I want to approach a topic I have been called out on several times. I believe there is a stigma that University researchers and extension specialists do not want products to work. It may seem that way at times but it is far from the truth. The reality is that all of us are scientists and know someone may be inventing the product that changes nutrient management as we speak. The issue is that most of us have been jaded. While I may be younger I have over 11 years experience, testing “products” in the field, and that includes dozens of products. I have sprayed, spread, tossed, drilled, mixed and applied everything under the sun, with hopes that I will see that one thing I am always looking for, MORE GRAIN…
The truth is Everything works Sometimes yet Nothing works ALL the time. I and others in my profession do not expect anything to work 100% of the time, I am personally looking for something that will provide a checkmark in the win column 50% of the time. A win is the result of one of two things, more money in the producers pocket or less nutrients in the water or air. Products can increase vigor, nutrient uptake, chlorophyll concentration, greenness but not yield. What Co-op or elevator pays for any of those attributes? Grain makes green.
So many safeners, stabilizers, enhancers, biologicals, and on and on are available, so what should a producer do? Here are few things to think about. Ask yourself “ what part of my nutrient management plan can I get the most bang from improving”?
If the answer is Nitrogen (N) there are three basic categories: Urease inhibition, Nitrification inhibitor, and slow release. All are methods of preventing loss; the last two are preventing loss from water movement.
Urease inhibitors prevent the conversion of Urea to NH3 (ammonia). This conversion is typically a good thing, unless it happens out in the open. Ideally any urea containing product is incorporated with tillage or rain. However, in No-till when urea is broadcast and no significant rainfall events (>0.5”) occur, N loss is likely. The urea prill starts dissolving in the presence of moisture, this can be a light rain or dew, and urease starts converting urea into NH3. As the system dries and the day warms, if there was not enough moisture to move the NH3 into the soil the wind will drive NH3 into the atmosphere. Nitrogen loss via this pathway can range from 5% to 40% of the total N applied.

Urea placed on a wet soil under two different temperatures. Number in white is hours after application.

Urea placed on a dry soil, on top no water added, bottom left is moisture from the subsurface, and bottom right is simulated rain fall of 1/2″. Number in white is hours after application.
Nitrification inhibitors prevent the conversion of NH4 into NO3. Both are plant available N sources but NH4 is a positively charged compound that will form a bound with the negatively charged soil particles. Nitrate (NO3) is negatively charged and will flow with the water, in corn country that tends to be right down the tile drainage. Nitrate will also be converted to gasses under wet water logged soil conditions. Nitrate is lost in the presence of water, this means I do not typically recommend nitrification inhibitors for western OK, KS, TX dryland wheat producers.
Slow release N (SRN) comes in a range of forms: coated, long chain polymer, organic and many versions in each category. Again, water is the reason for the use of SRN sources. Slow release N whether coated or other have specific release patterns which are controlled by moisture, temperature and sometimes microbes. The release patterns of SRNS are not the same and may not work across crops and landscapes. For instance in Oklahoma the uptake pattern of nutrients for dryland corn in the North East is not that same as irrigated corn in the West. The little nuances in the growth pattern of a crop can make or break your SRN.
While N products have been on the market for decade’s phosphorus enhancers and stabilizers are relatively new, resulting in many of my peers holding back on providing recommendations until field trials could be conducted. At this point many of us do have a better understanding of what’s available and are able to provide our regional recommendations. Phosphorus products are not sold to prevent loss like their N counterparts; they are sold to make the applied P more available. On a scale of 1 to 10, P reactivity with other elements in the soil is a 9.9. If there is available Ca, Mg, Fe, or Al, phosphorus is reacting with it. In the southern Great Plains it is not uncommon for a soil to have 3,000-5,000 lbs of available Ca, a soil with a pH of 4, yes we have many of those, will have approximately 64,000 lbs of Al in the soil solution. That’s a lot of competition for your fertilizer P and for any substance that is trying to protect it.
I have been testing “biologicals” of all shapes and forms since 2003. While I have not hit any homeruns I have learned quite a bit. Many of these products originate from up north where the weather is kind and organic matter (OM) is high. Where I work the average OM is 0.75% and soil temp is brutal and unforgiving. Our soil does not have many reserves to release nor is it hospitable to foreign bodies.
Soil temperature for Stillwater OK under sod and bare soil conditions. Graph from http://www.Mesonet.org.
I hope you are still hanging on as this next topic is a bit of a soap box for me. Rate, Rate, Rate this aspect is missed both by producers and academia and it drives me crazy. If your crop is sufficient in any growth factor adding more will not increase yield. It goes back to Von Liebig’s LAW of the Minimum. I see too many research studies in which products are tested at optimum fertilization levels. This is just not a fair comparison. On the other hand, time and again I see producers sold on a product because they applied 30% less N or P and cut the same yield. If you let me hand pick 100 farms in Oklahoma I could reduce the N rate by 30% of the average and not lose a bushel on 75 of the farms. Why? Because the rate being used was above optimum in the first place, there is no magic just good agronomy. The list of products that increase the availability of nutrients is a mile long. Increasing nutrient availability is all well and good if you have a deficiency of one of those nutrients. If you don’t, well you have increased the availability of something you did not need in the first place.
University researchers and extension professionals seem to live and die by the statistics, and are told so regularly. We do rely upon the significant differences, LSD’s, and etc to help us understand the likely hood of a treatment causing an effect. However if I see a trend develop, or not develop, over time and landscape regardless of stats I will have no problem making recommendations. The stats help me when I do not have enough information (replications).
Too wrap up, have a goal. Do not just buy a product because of advertised promises or because a friend sells it. There is a right time and place for most of the things out there, but you need to know what that is and if it suits your needs. I also recommend turning to your local Extension office. We do our best to provide unbiased information in hopes of making your operation as sustainable as possible. If you are looking at making sizable investments do some reading, more than just Google. Testimonies are great but should but should not be enough to cut a check. Google Scholar www.google.com/scholar is a good resource for scientific pubs. I have done my best to put together a list of peer reviewed publications and their outcomes. To make the review work I had to be very general about outcome of the research. Either the product increased yield or decreased environmental losses or it had no impact. This was not easy as many of the papers summarize multiple studies. I did my best to make an unbiased recommendation but some could be argued. http://npk.okstate.edu/Trials/products/Product_Peer_Review.8-21-2014.pdf
Banding P as a Band-Aid for low-pH soils.
In the mid-1970s Dr. Robert Westerman banded 18-46-0 with wheat at planting in a low-pH soil near Haskel Ok. The impact was immediately evident. Soon after Oklahoma State University recommended the “Banding of Phosphate in Wheat: A Temporary Alternative to Liming” Figure 1. This method was a Band-Aid solution for the significant amount Oklahoma winter wheat production area which was either too far from a reliable lime source or under a short term lease contract.
Still today grain producers throughout the United States commonly farm a large percentage of land that is not their own. In the leasing process agreements can widely vary both on length of the lease and the amount of inputs that the land owner will pay. The wheat belt of Oklahoma is known for having large areas with low soil pH levels. A survey of soil samples submitted to the Oklahoma State University Soil, Water, and Forage Analytical Laboratory in 2011 under the winter wheat crop code showed 38% of the samples having a soil pH level below 5.5 (Figure 2). In Oklahoma short term leases with limited shared expenses have limited the access to agricultural lime for remediation of acidic soils. In the dry environment it may take up to one year before the lime applied has completely corrected the soil acidity problem. In a situation where the lease agreement is only for one to two years there may be no economic benefit for the producer to apply lime especially in regions where winter wheat average yields range from 20 to 40 bushel per acre. The current recommendation for winter wheat producers working on low-pH short term lease ground is to apply 30 lbs P2O5 ac-1 ( 65 lbs 18-46-0 ac-1) with the seed for grain only wheat and 60 lbs P2O5 ac-1 (130 lbs 18-46-0 ac-1) for dual-purpose wheat production. This recommendation however is for soils with adequate soil test P, but low soil pH. When soil test P is below optimum the 30 or 60 units is applied in addition to the amount needed to reach 100% sufficiency.
Banding P is considered a “Band-Aid” as the problem of soil acidity is not re-mediated it is only masked. If not addressed the pH of the soil will continue to fall over time. Aluminum and manganese toxicity is the greatest issue associated with soil acidity. Available aluminum, a predominant mineral in the regions soils, is pH dependent. A change of 1.0 pH level changes available Al by 1000 fold. For example a soil with a pH of 5.0 will have an approximate Al concentration of 27 ppm, critical level of winter wheat is 27 ppm, while a soil with a pH of 4.0 will have an Al concentration of approximately 27,000 ppm. Aluminum and manganese toxicity does not only impact grain yield but it has an even greater impact on biomass production. Kariuki et al (2007) recorded the impact of soil acidity on eight current winter wheat lines. Correcting soil acidity increased wheat grain yield by 82% and increased forage production by 150%. For Oklahoma the forage produced by the wheat crop is as important as the grain. Oklahoma is unique in that approximately 50% of the four million acres of winter wheat are grazed annually much of this under the dual –purpose “Graze-N-Grain” management. To maintain productivity on the land without the long term investment of Ag lime producers have been applying phosphorus fertilizer to alleviate the impact of aluminum toxicity.

Figure 2. Summary of the soil pH values for the 614 samples submitted to the Oklahoma State University Soil, Water, Forage, Analytical Laboratory under wheat crop code during the time frame of 1-1-2011 11-30-2011.
In 1992 Boman et al reported that impact banding phosphates with seed on winter wheat forage production (Figure 3). Across the four locations the addition of P increase yield from 2 to 4 fold. The work by Kaitibie et al (2002) documented an additional aspect of banding P. In the variable and often arid climate of Oklahoma the activation of lime can take a significant amount of time, in upwards of one year. In comparison banding P has an immediate impact on the alleviation of metal toxicities. Figure 4 shows the incorporation of lime improved forage yield but not to the degree of banding P. For continuous winter wheat producers the time between application of lime and planting can be quite short. Typically the previous crop will be harvest in mid-June and in the best case scenario lime would be applied and incorporated by mid-July. At this point there is only 60 days until the next wheat crop is planted in early to mid-September.

Figure 3. The impact of banding phosphate with seed at planting in acidic soils on winter wheat forage production in Oklahoma. Chart adapted from Boman et al. 1992.

Figure 4. The impact of applying of phosphate fertilizer and lime on the forage production of two winter wheat cultivars in Oklahoma. Chart adapted from Kaitibie et al. 2002.
For many with short term leases banding P is still the only viable solution for wheat production in low-pH soils. However there is ground being farmed by the owner or is under long-term lease that is still receiving this Band-Aid approach. At the 1980-1990 fertilizer and lime prices there is good reason to continue this method. However the cost of P fertilizer has quadrupled since the 1970’s. The last ten year average price of P2O5 was $0.42 per pound while it cost an average of $0.10 in the 70s. So for those who own or are able to work out beneficial lease agreements Table 1 should be of interest. By year three the cost of phosphate exceeds the cost of lime. If you were to use the values from the 1980’s of $0.20 per pound of P2O5 and $25 per ton ECCE lime it was not until year five, the last year before reapplying lime, did the cost of P exceed cost of lime.

Table 1. Cumulative cost per acre of applying phosphorus and lime to remediate aluminum and manganese toxicity based on a five year liming cycle. The 30 lb P2O5 rate is recommended for grain only production while dual-purpose wheat require 60 P2O5. Prices based on current quotes of DAP at $590 a ton ($0.41 lb P2O5) and Ag lime at $30 per ton ECCE.
As the 2014 winter wheat and canola crop is being transported to the bins it is extremely important to take advantage of this time to take soil samples from as many fields as possible. Soil pH issues must be understood and addressed. I often remind producers soil pH plays an exception number of roles. Not only does it impact yield as shown before but it impacts rooting (ability to survive stresses), nutrient availability, and herbicide activity. Our SU herbicides (Finesse, Powerflex, and Maverick) that are used widely across the state are negatively impacted by low soil pH. Figure 5 shows how at a pH of 5.6 Glean is down to a 50% concentration in the soil approximately two weeks after application. So when it comes time to make the call for phosphorus or lime try to weigh all of these aspects, at current prices P is not that much cheaper, improving pH will improve yield and potentially improve weed control.

Figure 5. The concentration of Glean (Cholorsulfuron) remaining in too soils (pH 7.5 and pH 5.6) over a twelve week period.
Citations
Boman,R.K., R.L. Westerman, G.V. Johnson, and M.E. Jojola. 1992. Phosphorus fertilization effects on winter wheat production in acid soils. In Soil Fertility Highlights, Agronomy Department Oklahoma Agricultural Experiment Station, Oklahoma State University.Agronomy 92-1 pg171-174
Kaitibie,S., F. M. Epplin, E.G. Krenzer, and H. Zhang. 2002. Economics of lime and phosphorus application for dual-purpose wheat production in low-pH soils. Agron. J. 94:1139:1145.
Kariuki, S.K., H. Zhang, J.L. Schroder, J. Edwards, M. Payton, B.F. Carver, W.R. Raun, and E.G. Krenzer. 2007. Hard red winter wheat cultivar responses to a pH and aluminum concentration gradient. Agron J. 99:88-98.






