Home » sorghum
Category Archives: sorghum
Protect Your Emerging Stands: True Armyworm Movement from Maturing Wheat to Summer Crops
Ashleigh M. Faris, Cropping Systems Extension Entomologist & IPM Coordinator
As the Oklahoma winter wheat crop reaches maturity, producers and crop consultants should prepare for the annual migration of true armyworm larvae. While true armyworms are a common fixture in small grains, their movement out of maturing wheat and into newly emerged corn, soybeans, and sorghum can lead to stand thinning or loss if not monitored closely.
True Armyworm Migration Timeline
True armyworm moths typically migrate into Oklahoma from the south in early spring with infestations typically occurring in late April through the first two weeks of May. The first generation is typically laid in winter wheat. Once the larvae currently finish their development in wheat, they will soon seek new food sources as the wheat crop dries down. This transition period is the most critical time for scouting summer crops, especially those adjacent to wheat fields.
True Armyworm Life Cycle and Identification
Armyworms overwinter as pupae or as mature larvae which pupate in the spring. Moths emerge in the spring, mate, and lay eggs in masses on hosts plants (mostly in the grass family). Female moths deposit their eggs in low-lying areas on wheat or pasture ground, as well as field margins or fields with dense, grassy weeds like Johnson grass. Larvae feed for about 4 weeks but do most of their damage during the last 10 days of this period. They then pupate in the soil. A new generation of moths emerges about 1 week later. There are 4 generations per year in Oklahoma.
True armyworms have a smooth body and can be variable in color, ranging from green, tan, orange, and black, with distinct pale orange or reddish stripes running along the sides (Figure 1). A key identifier is a dark diagonal band on each of the abdominal prolegs; there are four pairs of prolegs (Figure 2). The head capsule is light brown with a distinct “net-like” or honeycomb pattern of dark lines (Figure 2).

Figure 1. Four true armyworm larvae. One is dark (right) and three are light colored (left). Photo by Ashley Dean, Iowa State University Extension.

Figure 2. True armyworm. A) Dark band on prolegs. B) Orange head capsule with dark net-like pattern. Photos by Adam Varenhorst, Iowa State University Extension.
True Armyworm Management Cutoff in Wheat
A common question during this window is whether to treat armyworms in maturing wheat. Once wheat reaches the soft dough stage, the crop has generally accumulated its yield. Unless larvae are actively head-clipping (cutting the wheat heads off the stems), chemical control is rarely economical at this stage. Instead of treating the wheat, focus on young stands of summer crops. As wheat turns brown, larvae will move toward the nearest green tissue—often your emerging corn or sorghum.
Scouting, Damage, and Economic Thresholds for Summer Crops
Armyworms are whorl feeders in grass crops like corn and sorghum and will also feed on soybean leaves. True armyworms hide in the soil, crop residue, or whorls during the heat of the day and feed at in the early morning, evening or late when it is cool outside. When it is warm, larvae will hide in the soil, crop residue, or the whorl of corn plants. Large larvae consume more tissue but will generally be done feeding in a few days. Insecticides should target young, small larvae that will be feeding for a long time; however, you may see a range of larval sizes in a single field.
Corn, Sorghum, and Soybean Damage
True armyworm feeding typically begins at the leaf edges, leaving ragged holes and edges (Figure 3). As this leaf tissue is removed, the larvae will move to the upper leaves and continue feeding. True armyworms do not tunnel into the stalk and generally do not feed on the growing point of larger corn and sorghum plants. While not the preferred host, true armyworms will move into soybeans if no grasses are available. Larvae typically cause defoliation (Figure 4); however, soybeans are quite resilient to early-season leaf loss, but scout for stand-thinning if larvae are clipping seedlings.

Figure 3. True armyworm feeding on young corn plant. Photo by Adam Varenhorst, Iowa State University Extension.

Figure 4. Soybean leaves with true armyworm feeding damage. Photo by Meaghan Anderson, Iowa State University Extension.
Corn Threshold: Small plants typically recover from true armyworm feeding and outgrow the defoliation. Per Kansas State Extension, treatment is justified only when larvae are less than 1.25 inches long and present on 30% of plants with 5 – 6 extended leaves, or when 75% of plants have one or more larva per plant. There is risk of yield loss if defoliation during reproductive stages approaches the ear zone before hard dent. Lower thresholds may apply if the plants are subject to additional stresses.
Sorghum Threshold: Sorghum is very tolerant of defoliation, so insecticide control is rarely justified. For early infestations (5-7 leaf stage, prior to panicle development) at the vegetative stages where true armyworms may be in the whorl, do not initiate controls unless 40% or more of the plants in a field are infested. Because the worms are only defoliating at this point in the sorghum plant’s development, economic damage is not a concern and there would likely be no return on investment for spraying before panicle development.
Soybean Threshold: Once grasses are fed upon or harvested, true armyworms can turn tobroadleaf crops, including soybean. While soybean is not a preferred host, the growing point is exposed early in the season, making them susceptible to stand loss. Management is suggested if soybean defoliation is greater than 35% – 40% during the vegetative stages.
True Armyworm Insecticide Management Options for Summer Crops
True armyworm is generally easier to control with pyrethroids than fall armyworm. Ensure high-volume water (10-15 GPA ground) is used to get the product into the whorl or canopy where the larvae hide. Remember that most insecticides work via contact; if true armyworm larvae are feeding or hiding under dense residue, insecticides are unlikely to make contact and are ineffective. Target applications when larvae are actively feeding on foliage to ensure good contact. Follow all instructions on the insecticide label to ensure good control.
For a complete list of recommended insecticides and rates for these crops, please consult the following OSU Fact Sheets: CR-7167: Management of Insect and Mite Pests in Corn and Sorghum and CR-7115: Management of Insect and Mite Pests in Soybean.
The information given herein is for educational purposes only. Reference to commercial products or trade names is made with the understanding that no discrimination is intended and no endorsement by the Cooperative Extension Service is implied.
Mechanisms of Soil Fertility: Looking at Biologicals and MOA
Brian Arnall, Oklahoma State University Precision Nutrient Management
The use of biological products in commercial agriculture has expanded rapidly, with large corporations entering a space once dominated by smaller groups. This has created an arms race, with nearly every company offering a biological product. Over the past twenty years, I have had the opportunity to test products from the biggest groups with billions in backing, to solutions raised in stock tanks delivered in Braums milk jugs. It is critical to understand what is in the jug and the biological function it is expected to perform. Like herbicides, knowing the mode of action determines whether the product fits the intended purpose. No different than herbicides and knowing mode of actions. It’s important to know and understand that if you are trying to kill ryegrass 2.4-D, a broadleaf herbicide is not the right answer.
So what are we working with that’s in these products?
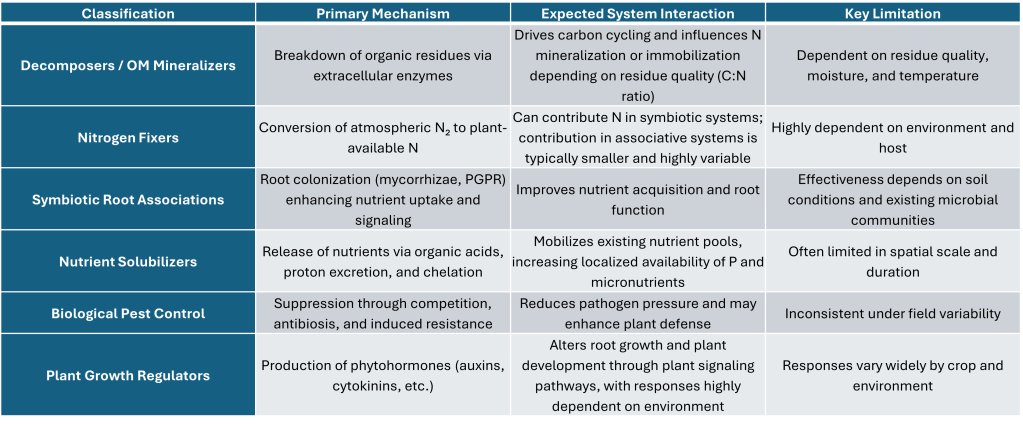
My approach has been to classify the products by operation not by species or genre. Doing so I have grouped products into five biological classifications and a sixth group, which is often in concluded in conversations.
Decomposers / Organic Matter Mineralizers
Nitrogen Fixers (Symbiotic and Associative)
Symbiotic Root Associations (Mycorrhizae, PGPR)
Nutrient Solubilizers
Biological Pest Control
Plant Growth Regulators (Hormonal Effects)
So, let’s dig into each of the mechanisms.
Decomposers / Organic Matter Mineralizers
Decomposition is carried out by a diverse group of organisms including fungi (e.g., Trichoderma, Aspergillus), bacteria (e.g., Bacillus, Pseudomonas), and actinomycetes (e.g., Streptomyces), each contributing to the breakdown of organic materials through different enzymatic pathways. This process of decomposing organic matter releases the nutrients tied up into plant available forms. The release of nitrogen is usually first thought, but this process adds significant amounts of potassium, calcium, and magnesium.
The process occurs both in the soil and on the soil surface. While it seems simple in application though this is a complex process. Let’s start with the soil pool, triggering decomposition of a system where the previous crop was wheat is significantly different than following corn. Following wheat, the carbon nitrogen ratio will be very high (see sugar blog), so while decomposition will release cations such as potassium and calcium, it is very likely to immobilize and residual nitrogen in the system. However, in fields that previously had corn the carbon to nitrogen ratio is much closer and the probability of seeing nitrogen release is much higher (Kuzyakov & Blagodatskaya, 2015). The process is similar for surface residues, but the rate is heavily controlled by rainfall. While both the soil and surface systems require moisture for the process to progress, the surface moisture is much more dynamic with frequent wetting and drying. Rain or irrigation is also needed to move the nutrients into the root zone.
One aspect of increasing decomposition of OM that I do not have a handle on is the long-term impact of expediting OM breakdown in and on the soil, especially in the central plains. As mentioned in the sugar blog, you would hope that the increase in nutrients from OM decomposition would increase plant growth enough to replenish the OM that was burned up. One caveat to this is that the decomposition would have to add nutrients that are deficient. Otherwise, there is no increase in plant growth and hypothetically the system is not net negative on OM. When it comes to decomposing surface residue, I have always been a bit hesitant in Oklahoma as I see having surface coverage to preserve soil moisture typically has a greater value than the nutrients from the residue.
Nitrogen Fixers (Symbiotic and Associative)
Nitrogen fixation is carried out by both symbiotic organisms such as Rhizobium and Bradyrhizobium, which form nodules on plant roots and supply significant nitrogen, and associative organisms such as Azospirillum and Azotobacter, which reside in the rhizosphere and contribute smaller, more variable amounts of nitrogen. Symbiotic nitrogen fixation, such as we have come to expect from legumes, is tightly regulated by the plant, with carbon supplied to the microbe in exchange for fixed nitrogen, making it one of the most efficient biological nitrogen inputs in agriculture.
Associative nitrogen fixation is not directly coupled to plant demand, and nitrogen contributions are typically limited by carbon availability and environmental conditions (Kennedy et al., 2004). While these organisms possess the ability to fix atmospheric nitrogen, the magnitude of nitrogen contribution, particularly from non-symbiotic systems, is highly variable and often limited under field conditions. We know that in soybean nodulation is greatly reduced when excess nitrogen is present in the soil, basically the plant does not need rhizobia, so it does not trigger symbiosis. I expect that as we move symbiotic fixation out of legumes that this mechanism does not change. Finally fixed N is no different than fertilizer N, if you add more then the crop needs, its lost. Therefore, if I am planning to use a N fixer, I would significantly reduce the amount of fertilizer N apply well below crop demand. Otherwise, the money spent on the N fixer is a waste. The only argument I have heard for this is the security blanket, making sure that if more is needed than normally the system is covered. But I circle back to the question about a system with high levels of residual N and rhizobium nodulation.
Symbiotic Root Associations (Mycorrhizae, PGPR)
Symbiotic root associations include arbuscular mycorrhizal fungi (e.g., Rhizophagus, Funneliformis) that extend the effective root system and improve nutrient uptake, particularly phosphorus, as well as plant growth-promoting rhizobacteria (e.g., Pseudomonas, Bacillus) that influence root development and plant signaling through multiple biochemical pathways (Smith & Read, 2008). In my visits with soil microbiologist, I have been left with the understanding that these relationships are not generic, but quite specific. There is significant influence of genotype and environment. And even more interesting is that the majority expect that the plant needs to signal for this relationship to happen.
The effectiveness of these associations is highly dependent on soil conditions, existing microbial communities, and nutrient availability, with responses often diminishing in systems where nutrients are not limited or where native populations are already established. I was able to follow along with some work down at OSU a few years back that was working with sorghum looking for symbiotic relationships to improve water and nutrient uptake specifically phosphorus. The work was successful, the researchers were able to identify a AMF that created a symbiotic relationship with sorghum, with a few caveats. First land race cultivars had a much higher incidence of symbiosis. For the landraces it worked well in extremely nutrient depleted soils and any additions of N or P reduced forage yield over the none. In the end the researchers were able to show improved the grain yield in landraces above fertilized, but these yields did equal fertilized hybrids. This work had great impact on small holders in developing counties with limited resources.
Nutrient Solubilizers
Nutrient solubilization is carried out by organisms such as Bacillus, Pseudomonas, and Aspergillus, which increase nutrient availability through mechanisms including organic acid production, proton release, and chelation, allowing nutrients like phosphorus and micronutrients to become more accessible in the rhizosphere.
Phosphorus-solubilizing fungi, such as Aspergillus and Penicillium, function similarly to bacterial solubilizers but are often more effective at producing strong organic acids. These acids can lower pH in localized zones and release phosphorus from mineral-bound forms, particularly in soils with high fixation capacity. Fungal systems can operate across a wider range of environmental conditions and may play a larger role in longer-term phosphorus cycling. However, as with bacterial systems, these effects are generally localized and dependent on soil chemistry (Richardson et al., 2009). I tend to see these having the greatest benefits in systems that have historically received manures or long-term applications of fertilizer P. I do not believe this is a good fit for soils with limited available phosphorus, as it is trying to focus the soil into something, it does not want to do or have too spare.
Potassium-solubilizing organisms, including species such as Bacillus mucilaginosus and Frateuria aurantia, contribute to the release of potassium from primary minerals like feldspars and micas. These microbes facilitate mineral weathering through acidification and chelation processes that slowly break down mineral structures. While the mechanism is well understood, the rate of potassium release is typically slow relative to crop demand. As a result, these organisms are more influential in long-term soil development than in short-term fertility management (Sheng & He, 2006).
Micronutrient-mobilizing organisms, particularly Pseudomonas and Bacillus species, enhance availability through the production of siderophores and other chelating compounds. These molecules bind metals such as iron and zinc, increasing their solubility and facilitating uptake in the rhizosphere. This process is especially important in soils where micronutrients are present but not readily available due to chemical constraints. However, the impact is typically limited to the immediate root zone and depends on both microbial activity and soil conditions (Ahmed & Holmström, 2014).
Biological Pest Control
Biological pest control organisms, including species such as Bacillus, Pseudomonas, and Trichoderma, function by suppressing pathogens through several well-documented mechanisms. These include the production of inhibitory compounds, competition for space and nutrients, direct antagonism of pathogens, and the activation of plant defense systems through induced systemic resistance. While these mechanisms are well established under controlled conditions, their effectiveness in field environments is highly dependent on environmental conditions, pathogen pressure, and the ability of the organism to persist and colonize the soil or plant surface (Lugtenberg & Kamilova, 2009).
I’ve been working with a lot of folks from Brazil who historically make four to six nemacide applications in soybean, but utilizing Pseudomanas they have been able to reduce that number by half or more. The caveat, as I understand, the application rates needed are significantly higher than anything I have seen in the US. If you look through the literature, you are seeing more and more documentation of this such as Li et al. 2022. But as Spescha et al. (2023) documented, different biological control agents operate through complementary mechanisms, including infection, toxin production, and host targeting. However, effectiveness depended on environmental conditions and interactions among organisms, reinforcing that biological control outcomes are system-dependent rather than universally consistent.
Plant Growth Regulators (Hormonal Effects)
This group differs slightly, as the primary effect is not direct nutrient cycling but modification of plant physiological response. This group is one I hold the greatest expectations for. I mean we have been using PGRs in crop production for decades, we just did not have an inkling of how many PGRs exist.
Plant growth regulator effects are associated with organisms such as Azospirillum, Bacillus, and Pseudomonas, which can influence plant development through the production of phytohormones and related compounds. These microbes produce substances such as auxins, cytokinins, and gibberellins that alter root architecture and plant growth patterns, and in some cases reduce stress responses through enzymes like ACC deaminase. Rather than supplying nutrients directly, these organisms modify how plants respond to their environment and utilize available resources. However, just like everything previously discussed the magnitude of response is often subtle and highly dependent on environmental conditions and crop system interactions (Glick, 2012).
Final thoughts.
There is one situation that pops up that I do not agree with, based upon my limited understanding of soil microbiology. Its adding more of what is already there. The soil system is a dynamic system. While there are population booms and bust, it supports what it is able to. Adding more of what is already there is like dropping a million rabbits into a prairie that has rabbits already. The current population is where it is because that is what the system can support. Adding means one of two things, a lot of rabbits die immediately, or they overwhelm the system and another animal species dies off due to lack of resources. Also, most microbiologists tell me the system is amazing at signaling and finding what it wants. It may take a season, but it will be there, in the quantities that soil needs, just given time.
So, the final slide in all my biological additives talks ends with this statement. My experiments show one thing. The impact of adding these products on crop yields is very consistently inconsistent. I’ve had many show a significant positive response, once. I have struggled to ever get repeated successes. It is my belief that I will have more success improving the soil biome by managing the soil (no-till, crop rotation, cover crops) than I will ever have with adding a product.
Final comment, Read the label. Many of the biological products I have tested are not singularly pure species. There are many blends of species and organisms which encompass many of the modes. A lot of these blends also contain extras such as humics, fulvics, carbohydrates, and sugars, see previous blogs.
Take-Home Messages
- Biological products function through specific mechanisms, not as broad “boosters,” and understanding that mechanism is critical to proper use.
- The presence of a biological function does not guarantee a yield response, outcomes are driven by soil, crop, and environmental conditions
- Decomposers and carbon-driven systems can immobilize or mineralize nitrogen, depending largely on residue quality and system balance
- Mycorrhizae and PGPR improve access to existing nutrients, not total nutrient supply
- Nutrient-solubilizing organisms mobilize nutrients already present in the soil
- Plant growth regulators influence plant signaling and development
- Adding biological organisms to soil does not guarantee establishment or persistence, as soil systems can regulate microbial populations.
- Management practices such as no-till, crop rotation, and cover crops are effective at improving soil biological function
- Across all biological products, mechanism exists, but response depends on the system
Any questions or comments please reachout to me @ b.arnall@okstate.edu
Citations
Ahmed, E., & Holmström, S. J. M. (2014). Siderophores in environmental research: Roles and applications. Microbial Biotechnology, 7(3), 196–208.
Glick, B. R. (2012). Plant growth-promoting bacteria: Mechanisms and applications. Scientifica, 2012, 963401
Kennedy, I. R., Choudhury, A. T. M. A., & Kecskés, M. L. (2004).
Non-symbiotic bacterial diazotrophs in crop-farming systems. Plant and Soil, 266, 65–79.
Kuzyakov, Y., & Blagodatskaya, E. (2015).
Microbial hotspots and hot moments in soil. Soil Biology and Biochemistry, 83, 184–199.
Lugtenberg, B., & Kamilova, F. (2009). Plant-growth-promoting rhizobacteria. Annual Review of Microbiology, 63, 541–556.
Richardson, A. E., Barea, J. M., McNeill, A. M., & Prigent-Combaret, C. (2009). Acquisition of phosphorus and nitrogen in the rhizosphere and plant growth promotion by microorganisms. Plant and Soil, 321(1–2), 305–339.
Sheng, X. F., & He, L. Y. (2006). Solubilization of potassium-bearing minerals by a wild-type strain of Bacillus edaphicus and its mutants and increased potassium uptake by wheat. Canadian Journal of Microbiology, 52(1), 66–72. https://doi.org/10.1139/w05-117
Smith, S. E., & Read, D. J. (2008).
Mycorrhizal symbiosis. Academic Press.
Spescha, A., Weibel, J., Wyser, L., Brunner, M., Hess Hermida, M., Moix, A., Scheibler, F., Guyer, A., Campos-Herrera, R., Grabenweger, G., & Maurhofer, M. (2023). Combining entomopathogenic Pseudomonas bacteria, nematodes and fungi for biological control of a below-ground insect pest. Agriculture, Ecosystems & Environment, 348, 108414.
Ye S, Yan R, Li X, Lin Y, Yang Z, Ma Y and Ding Z (2022) Biocontrol potential of Pseudomonas rhodesiae GC-7 against the root-knot nematode Meloidogyne graminicola through both antagonistic effects and induced plant resistance. Front. Microbiol. 13:1025727. doi: 10.3389/fmicb.2022.1025727
The Mechanics of Soil Fertility: Use of Sugar in Field Crops
Jolee Derrick, Precision Nutrient Management Ph. D. Student
Grace Williams, Soil Microbiology Ph. D. Candidate
Brian Arnall, Precision Nutrient Management Specialist
Recently, there has been increased interest in adding sugar to spray tank mixes, whether for post-emergence weed control or foliar nutrient applications. While there is limited work on impact of sugar inclusion in herbicide applications, some papers have posed potential enhancement (Devine and Hall, 1990). But since this is coming from a soil science group, we will only focus on soil impact. Following up the last blog, unlike humic substances, which represent more complex and relatively stable carbon forms, sugar is a highly labile carbon source. This rapid utilization of simple carbon sources is well documented to stimulate microbial activity and growth (Kuzyakov and Blagodatskaya, 2015). The general idea of utilizing sugar applications is that sugar has the capacity to improve spray performance, stimulate biological activity, increase organic matter mineralization, and ultimately result in improved yields.
Sugar additions can influence soil processes differently depending on system conditions. In systems with higher residual nitrogen and organic matter, responses may differ from those observed in Oklahoma production environments, where soils are typically lower in organic matter and microbial activity can occur for much of the year. Understanding how sugar functions in these systems requires a basic discussion of carbon dynamics. Sugar itself is almost entirely carbon and is readily consumed by microbes. It’s a simple molecule, which allows it to dissolve easily in water and be quickly utilized in the soil system. Crop residues, like wheat straw, are also carbon-rich but much more complex. They contain cellulose, hemicellulose, and lignin which are long carbon chains that take time to break down because microbes need specialized enzymes to access them.
For the sake of simplicity, we can group carbon into two key pools: labile carbon and particulate organic matter (POM). Labile carbon includes easily decomposed materials, which include the previously mentioned simple sugars that microbes can metabolize rapidly. These pools differ in turnover time and microbial accessibility, with labile carbon driving short-term microbial responses (Cotrufo et al. 2013). POM breaks down more slowly and serves as a longer-term nitrogen source through residue breakdown.
Soil microorganisms require both carbon and nitrogen to grow and maintain biomass, typically at a ratio of approximately 24 parts carbon to 1 part nitrogen. When readily available carbon is abundant, but nitrogen is limited, microbes increase their nitrogen demand and begin scavenging nitrogen from the surrounding soil. This process, better known as nitrogen immobilization, temporarily reduces nitrogen availability to crops. Additions of readily available carbon sources have consistently been shown to increase microbial nitrogen immobilization in soil systems (Recous et al. 1990).
In systems where sufficient nitrogen is present, microbial populations can expand rapidly. Fast-growing microbial species may dominate, continuing to immobilize nitrogen within their biomass. Eventually, when nitrogen becomes limiting, microbial populations decline to levels the system can support. This boom-and-bust cycle can disrupt nitrogen availability during critical stages of crop growth. These rapid shifts in microbial population and activity following carbon inputs are commonly observed in soil systems receiving easily decomposable substrates (Blagodatskaya and Kuzyakov, 2008).
This dynamic becomes especially relevant when considering residue management practices common in Oklahoma. Under no-till or limited-tillage systems, the crop residues have wide carbon-to-nitrogen (C:N) ratios, creating conditions where nitrogen immobilization can occur during the growing season.
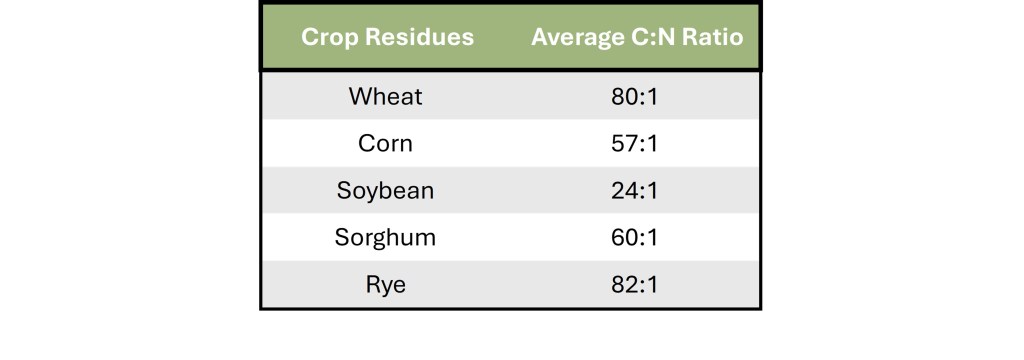
Table 1 provides approximate C:N ratios for several crops commonly grown in Oklahoma. When additional carbon is introduced into these systems without accompanying nitrogen, the likelihood of microbial immobilization increases. While immobilization is not bad, it does create a question mark as Oklahoma’s variable climate means the following release of nutrients will be unpredictable.
Table 1. Table depicting the range of C:N ratios for residues of commonly utilized crops in Oklahoma. Ratios were obtained from Brady, N. C., & Weil, R. R. (2017). The Nature and Properties of Soils (15th ed.)
Now consider conventional tillage systems. In Oklahoma, no-till systems typically contain 2 to 3 percent organic matter, which is relatively high given our climate and extended periods of microbial activity. Conventional tillage systems often fall between 0.75 and 2.25 percent organic matter. Because soil organic matter is approximately 58 percent carbon, this represents a substantial difference in the soil carbon pool.
Tillage can temporarily enhance microbial access to both previously mentioned carbon pools. When tillage exposes previously protected carbon, microbial activity increases rapidly. This initial flush can temporarily increase nitrogen mineralization as organic nitrogen is converted to plant-available forms. However, this phase is short-lived. As microbial populations expand, nitrogen demand increases, leading to immobilization and reduced nitrogen availability.
Hypothetically, increased microbial growth and activity would rapidly mineralize organic matter, trigger a surge in NO₃⁻, deplete soil organic matter, and as resources become limiting and the environment can no longer sustain elevated microbial populations, this boom would be followed by a population crash. This relationship is ultimately driven by the soil C:N ratio, which introduces an interesting additional complexity of residue. Different residues bring very different carbon-to-nitrogen balances into the system, and microbes respond accordingly. High carbon residues give microbes plenty of energy but very little nitrogen, so they pull N out of the soil to meet their needs. Residues with lower C:N ratios (soybean, alfalfa, etc.) do opposite, releasing nitrogen as they break down. Now the real question becomes where the critical point sits, and when does management push the system from the threshold of immobilization and mineralization.
These hypotheses form the foundation for new research currently underway through the Precision Nutrient Management Program. Initial proof-of-concept work has already been completed, providing a necessary steppingstone to address these questions.
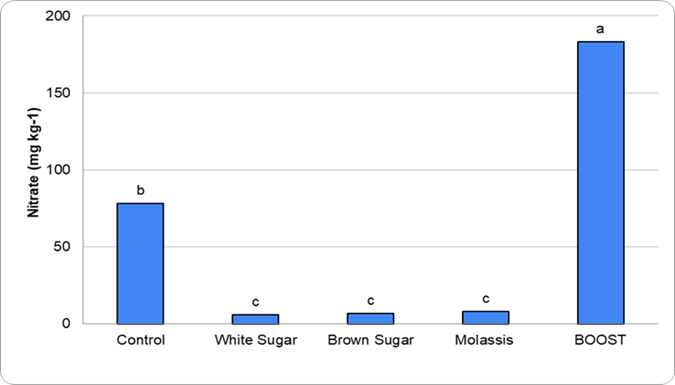
Figure 1. Graph depicting the different concentrations of nitrate leached corresponding to applied treatments in the proof-of-concept work
The preliminary work (Figure 1) evaluated different sugar sources applied alongside a high-nitrogen product to assess the extent of nitrogen immobilization. Although these studies were conducted using potting soils, clear trends were apparent. Treatments containing sugar consistently showed greater nitrogen immobilization compared to treatments without sugar. This response is consistent with studies showing that additions of simple carbon substrates stimulate microbial growth and increase nitrogen immobilization (Dendooven et al. 2006). Building on this work, an active field-based research project is underway to evaluate how sugar additions influence nitrogen availability and microbial dynamics under real-world Oklahoma production conditions.
From an agronomic standpoint, sugar functions primarily as a readily available carbon source that stimulates microbial growth. In nitrogen-limited systems, this response increases the likelihood that nitrogen will be incorporated into microbial biomass rather than remaining immediately available for crop uptake.
Finally, we conclude with a conceptual consideration. If increased OM mineralization leads to greater plant biomass, this process may partially offset losses of OM. Greater biomass production could return more residues to the soil, contributing to the OM pool in the upper soil profile. Therefore, the system may compensate for OM mineralization through the rebuilding of organic matter via plant inputs. However, the stabilization of this carbon depends on microbial processing and physical protection within the soil matrix (Cotrufo et al. 2015)
However, while the underlying logic is sound, this concept has not been extensively studied within Oklahoma cropping systems. This blog does not address the impact of sugar applications on residue breakdown, and the potential impact of such. Future research through the Precision Nutrient Management Program will further investigate the mineralization process to better understand carbon dynamics within these systems.
Take Home:
- Oklahoma production systems generally have lower residual N and high carbon residues, creating conditions conducive to N immobilization
- Adding sugar increases microbial growth, creating population booms that will momentarily increase mineralization, but then immediately immobilize residual nitrogen.
- Tillage can amplify the negative effects of sugar by exposing more carbon and reducing soil organic matter
- Proof-of-concept work shows sugar triggered a net nitrogen immobilization in a carbon heavy environment
- Proof-of-concept work also suggests that when additional nitrogen is present, sugar additions may shift the system toward net mineralization rather than immobilization.
Work Cited:
Blagodatskaya, E., & Kuzyakov, Y. (2008). Mechanisms of real and apparent priming effects. Biology and Fertility of Soils, 45, 115–131.
Brady, N. C., and R. R. Weil. “The Nature and Properties of Soils, 15th Edn (eBook).” (2017).
Cotrufo, M. F., Wallenstein, M. D., Boot, C. M., Denef, K., & Paul, E. (2013). The Microbial Efficiency-Matrix Stabilization (MEMS) framework. Global Change Biology, 19, 988–995.
Cotrufo, M. F., Soong, J. L., Horton, A. J., Campbell, E. E., Haddix, M. L., Wall, D. H., & Parton, W. J. (2015). Formation of soil organic matter via biochemical and physical pathways of litter mass loss. Nature Geoscience, 8(10), 776–779.
Dendooven, L., Verhulst, N., Luna-Guido, M., & Ceballos-Ramírez, J. M. (2006). Dynamics of inorganic nitrogen in nitrate- and glucose-amended alkaline–saline soil. Plant and Soil, 283(1–2), 321–333.
Devine, M. D., & Hall, L. M. (1990). Implications of sucrose transport mechanisms for the translocation of herbicides. Weed Science, 38(3), 299–304.
Kuzyakov, Y., & Blagodatskaya, E. (2015). Microbial hotspots and hot moments in soil: Concept & review. Soil Biology and Biochemistry, 83, 184–199.
Recous, S., Mary, B., & Faurie, G. (1990). Microbial immobilization of ammonium and nitrate in cultivated soils. Soil Biology and Biochemistry, 22, 913–922.
Thoughts from an Agronomist- 1 Management of the Primordia
Josh Lofton, Cropping Systems Specialist
Many crop management recommendations emphasize actions that must be taken well before a crop reaches what we often call “critical growth stages.” Management this early can seem counterintuitive when the crop still looks small, healthy, or unchanged aboveground. However, much of a crop’s yield potential is determined early in the season at a level we cannot see in the field. Long before flowers, tassels, or heads (or any reproductive structure) appear, the plant is already making developmental decisions that shape its final yield potential. Understanding this “behind the scenes” process helps explain why timely, early-season management is often more effective than trying to correct problems later.
At the center of this process is the shoot apical meristem, commonly referred to as the growing point. This tissue produces leaf and reproductive primordia, which are the earliest developmental stages of future everything in the plant. These primordia form well before the corresponding plant parts are visible. Once these structures initiate—or if they fail to begin due to stress—the outcome is permanent. The plant cannot later in the season go back and recreate leaf number, leaf size, or reproductive capacity. As a result, early environmental conditions and management decisions play a disproportionate role in determining yield potential.
Corn is a good example of how early development influences final yield. By the time corn reaches the V4 growth stage, the plant only has four visible leaves with collars, yet internally it is far more advanced. Most of the total leaf primordia that will eventually form the full canopy have already begun, and the potential size of the ear is starting to be established. During this stage, the growing point is still below the soil surface and somewhat protected from some stressors but highly susceptible to others. Nitrogen deficiency, cold temperatures, moisture stress, compaction, or herbicide injury at or before V4 can reduce leaf number and limit leaf expansion. Even if growing conditions improve later, the plant cannot replace leaf primordia that were never formed, which reduces its ability to intercept sunlight and support high yields.

As corn approaches tasseling (VT), the crop enters a stage that is visually and physiologically important. Pollination, fertilization, and early kernel development occur at this time, and stress can have a critical impact on kernel set. However, by VT, the plant has already completed leaf formation, and much of the ear size potential has already been determined several growth stages earlier. Management at VT is therefore focused on protecting yield rather than creating it. Late-season nutrient applications may improve plant appearance or maintain green leaf area, but they cannot increase leaf number or rebuild ear potential lost due to early-season stress. This distinction helps explain why some late inputs show limited yield response even when the crop looks responsive.
Grain sorghum provides another clear example of why early management is emphasized. Although sorghum often grows slowly early in the season and may appear unimportant during the first few weeks after emergence, the first 30 days are among the most critical periods in its development. During this time, the growing point is actively producing leaf primordia and transitioning from vegetative growth toward reproductive development. Head size potential is primarily established during this early window, and the plant’s capacity to support tillers is influenced by early nutrient availability and moisture conditions. Stress from nitrogen deficiency, drought, weed competition, or restricted rooting during the first 30 days can reduce head size and kernel number long before visible symptoms appear.
Once sorghum reaches later vegetative and reproductive stages, much like corn at VT, management shifts from building yield potential to protecting what has already been determined. Improving conditions later in the season can help maintain plant health and grain fill, but it cannot fully compensate for early limitations imposed at the primordial level. This is why early fertility placement, timely weed control, and moisture conservation are consistently emphasized in sorghum production systems.
Across crops, a typical pattern emerges: the growth stages we observe in the field often reflect decisions the plant made weeks earlier. When agronomists stress early-season management, they are responding to plant biology rather than simply following tradition. By the time visible “critical stages” arrive, the plant has already established many of the components that define yield potential.
The key takeaway is that effective crop management must be proactive rather than reactive. Early-season decisions support the crop while it is still determining how many leaves it can produce, how large its reproductive structures can become, and how much yield it can ultimately support. Waiting until stress becomes visible often means responding after the plant has already adjusted its potential downward. Recognizing what is happening at the primordial level helps explain why management ahead of critical stages consistently delivers the greatest return, even when the crop appears small and unaffected aboveground.
For questions or comments reach out to Dr. Josh Lofton
josh.lofton@okstate.edu
Double Crop Options After Wheat (KSU Edition)
Stolen from the KSU e-Update June 5th 2025.
Double cropping after wheat harvest can be a high-risk venture for grain crops. The remaining growing season is relatively short. Hot and/or dry conditions in July and August may cause problems with germination, emergence, seed set, or grain fill. Ample soil moisture this year can aid in establishing a successful crop after wheat harvest. Double-cropping forages after wheat works well even in drier regions of the state.
The most common double crop grain options are soybean, sorghum, and sunflower. Other possibilities include summer annual forages and specialized crops such as proso millet or other short-season summer crops, even corn. Cover crops are also an option for planting after wheat (see the companion eUpdate article “Cover crops grown post-wheat for forage”).
Be aware of herbicide carryover potential
One major planting consideration after wheat is the potential for herbicide carryover. Many herbicides applied to wheat are Group 2 herbicides in the sulfonylurea family with the potential to remain in the soil after harvest. If a herbicide such as chlorsulfuron (Glean, Finesse, others) or metsulfuron (Ally) has been used, then the most tolerant double crop will be sulfonylurea-resistant varieties of soybean (STS, SR, Bolt) or other crops. When choosing to use herbicide-resistant varieties, be sure to match the resistance trait with the specific herbicide (not only the herbicide group) that you used. This is especially true when looking at sunflowers as a double crop. There are sunflowers with the Clearfield trait, which allows Beyond herbicide applications, and ExpressSun sunflowers, which allow an application of Express herbicide. While both of these herbicides are Group 2 (ALS-inhibiting herbicides), the Clearfield trait and ExpressSun are not interchangeable, and plant damage can result from other Group 2 herbicides.
Less information is available regarding the herbicide carryover potential of wheat herbicides to cover crops. There is little or no mention of rotational restrictions for specific cover crops on the labels of most herbicides. However, this does not mean there are no restrictions. Generally, there will be a statement that indicates “no other crops” should be planted for a specified amount of time, or that a bioassay must be conducted prior to planting the crop.
Burndown of summer annual weeds present at planting is essential for successful double-cropping. Assuming glyphosate-resistant kochia and pigweeds are present, combinations of glyphosate with products such as saflufenacil (Sharpen) or tiafenacil (Reviton), or alternative treatments such as paraquat may be required. Dicamba or 2,4-D may also be considered if the soybean varieties with appropriate herbicide resistance traits are planted. In addition, residual herbicides for the double crop should be applied at this time.
Management, production costs, and yield outlooks for double crop options are discussed below.
Soybeans
Soybeans are likely the most commonly used crop for double cropping, especially in central and eastern Kansas (Figure 1). With glyphosate-resistant varieties, often the only production cost for planting double crop soybeans was the seed, an application of glyphosate, and the fuel and equipment costs associated with planting, spraying, and harvesting. However, the spread of herbicide-resistant weeds means additional herbicides will be required to achieve acceptable control and minimize the risk of further development of resistant weeds.

Weed control. The weed control cost cannot really be counted against the soybeans, since that cost should occur whether or not a soybean crop is present. In fact, having soybeans on the field may reduce herbicide costs compared to leaving the field fallow. Still, it is recommended to apply a pre-emergence residual herbicide before or at planting time. Later in the summer, a healthy soybean canopy may suppress weeds enough that a late-summer post-emergence application may not be needed.
Variety selection for double cropping is important. Soybeans flower in response to a combination of temperature and day length, so shifting to an earlier-maturing variety when planting late in a double crop situation will result in very short plants with pods that are close to the ground. Planting a variety with the same or perhaps even slightly later maturity rating (compared to soybeans planted at a typical planting date) will allow the plant to develop a larger canopy before flowering. Planting a variety that is too much later in maturity, however, increases the risk that the beans may not mature before frost, especially if long periods of drought slow growth. The goal is to maximize the length of the growing season of the crop, so prompt planting after wheat harvest time is critical. The earlier you can plant, the higher the yield potential of the crop if moisture is not a limiting factor.
Fertilizer considerations. Adding some nitrogen (N) to double-crop soybeans may be beneficial if the previous wheat yield was high and the soil N was depleted. A soil test before wheat harvest for N levels is recommended. Use no more than 30 lbs/acre of N. It would be ideal to knife-in the fertilizer. If that is not possible, banding it on the soil surface would be acceptable. Do not apply N in the furrow with soybean seed as severe stand loss can occur.
Seeding rates and row spacing. Seeding rate can be slightly increased if soybeans are planted too late in order to increase canopy development. Narrow row spacing (15-inch or less) has often resulted in a yield advantage compared to 30-inch rows in late plantings. Soybeans planted in narrow rows will canopy over more quickly than in wide rows, which is important when the length of the growing season is shortened. Narrow rows also offer the benefits of increasing early-season light capture, suppressing weeds, and reducing erosion. On the other hand, the advantage of planting in wide rows is that the bottom pods will usually be slightly higher off the soil surface to aid harvest. The other consideration is planting equipment. Often, no-till planters will handle wheat residue better and place seeds more precisely than drills, although the difference has narrowed in recent years.
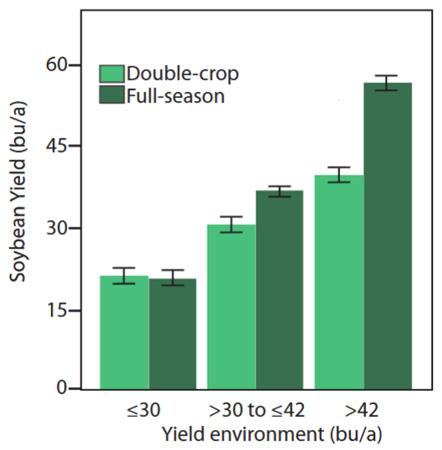
What are typical yield expectations for double-crop soybeans? It varies considerably depending on moisture and temperature, but yields are usually several bushels less than full-season soybeans. A long-term average of 20 bushels per acre is often mentioned when discussing double-crop soybeans in central and northeast Kansas. Rainfall amount and distribution can cause a wide variation in yields from year to year. Double-crop soybean yields typically are much better as you move farther southeast in Kansas, often ranging from 20 to 40 bushels per acre.
A recent publication explores the potential yield of double-crop soybeans relative to full-season yield (Figure 2) and the most limiting factors affecting the yields for double-crop soybeans. The link to this article is: https://bookstore.ksre.ksu.edu/pubs/MF3461.pdf.
Grain Sorghum
Grain sorghum is another double crop option. Unlike soybeans, sorghum hybrids for double cropping should be earlier maturing hybrids. Sorghum development is primarily driven by the accumulation of heat units, and the double crop growing season is too short to allow medium-late or late hybrids to mature before the first frost in most of Kansas.
Seeding rates and row spacing. Late-planted sorghum likely will not tiller as much as early plantings and can benefit from slightly higher seeding rates than would be used for sorghum planted at an earlier date. Narrow row spacing is advised, especially if the outlook for rainfall is good.
Fertilizer considerations. A key component for the estimation of N application rates is the yield potential. This will largely determine the N needs. It is also important to consider potential residual N from the wheat crop. This can be particularly important when wheat yields are lower than expected. In that situation, additional available N may be present in the soil. Assess the amount of profile N by taking soil samples at a depth of 24 inches and submitting them for analysis at a soil testing laboratory.
Double crop sorghum planted into average or greater-than-average amounts of wheat residue can result in a challenging amount of residue to deal with when planting next year’s crop. Nitrogen fertilizer can be tied up by wheat residue, so use application methods to minimize tie-up, such as knifing into the soil below the residue.
Weed control. Weed control can be important in double-crop sorghum. Warm-season annual grasses, such as crabgrass, can reduce double-crop sorghum yields. Using a chloroacetamide-and-atrazine pre-emergence product may be key to successful double-crop sorghum production. Herbicide-resistant grain sorghum varieties will allow the use of imazamox (Imiflex in igrowth sorghums) or quizalofop (FirstAct in DoubleTeam grain sorghum) that can control summer annual grasses.
No-till studies at Hesston documented 4-year average double crop sorghum yields of 75 bushels per acre compared to about 90 bushels per acre for full-season sorghum. A different 10-year study that did not have double crop planting but did compare early- and late-planting dates averaged 73 bushels per acre for May planting vs. 68 bushels per acre for June planting.
Sunflowers
Sunflowers can be a successful double crop option anywhere in the state, provided there is enough moisture at planting time to get a stand. Sunflowers need more moisture than any other crop to germinate and emerge because of the large seed. Therefore, stand establishment is important. Planting immediately after wheat harvest on a limited irrigation field can be a good fit to help with stand establishment.
Seeding rates and hybrid selection. When double-cropping sunflowers, producers should use similar seeding rates to what is typical for the area for full-season sunflowers. While full-season sunflowers can be successful in double-crop production, utilizing shorter-season hybrids can increase the likelihood of the sunflowers blooming and maturing before a killing frost.
Weed control. First, it is important to check the herbicide applications on the wheat. The rotation restriction to sunflowers after several commonly used wheat herbicides is 22-24 months.
Weed control can be an issue with double-crop sunflowers since herbicide options are limited, especially post-emergence. Thus, controlling weeds prior to sunflower planting is critical and may be complicated pre-plant restrictions for some herbicides. Planting Clearfield or ExpressSun sunflowers will provide additional post-emergence herbicide options, but ALS-resistant kochia and pigweeds still won’t be controlled. Imazamox (Beyond in Clearfield sunflower) has activity on small annual grasses as well as many broadleaf weeds, if they are not ALS-resistant.
Summer annual forages
With mid-July plantings, and where herbicide carryover issues are not a concern, summer annual sorghum-type forages are also a good double crop option. A study planted July 21, 2008 near Holton, when summer rainfall was very favorable, provided yields of 2.5 to 3 tons dry matter/acre for hybrid pearl millet and sudangrass at the low end to 4 to 5 tons dry matter/acre for forage sorghum, BMR forage sorghum, photoperiod sensitive forage sorghum, and sorghum x sudangrass hybrids. Earlier plantings may produce even more tonnage, as long as there is adequate August rainfall.
One challenge with late-planted summer annual forages is getting them to dry down when harvest is delayed until mid- to late-September. Wrapping bales or bagging to make silage are good ways to deal with the higher moisture forage this late in the year.
Corn
Is double-crop corn a viable option? Corn is typically not recommended for late June or July plantings because yield is usually substantially less than when planted earlier.
Typically, mid-July planted corn struggles during pollination and seldom receives sufficient heat units to fill grain before frost. Very short-season corn hybrids (80 to 95 RM) have the greatest chance of maturing before frost in double crop plantings, but generally have less yield potential when compared to hybrids of 100 RM or more used for full-season plantings. Short-season hybrids often set the ear fairly close to the ground, increasing the harvest difficulty. Glyphosate-resistant hybrids will make weed control easier with double crop corn, but problems remain present with late-emerging summer weeds such as pigweeds, velvetleaf, and large crabgrass. Keep in mind, corn is very susceptible to carryover of most residual ALS herbicides used in wheat.
Considerations for altering seeding rates and variety/hybrid maturity for the crops discussed above are summarized in Table 1.
Table 1. Seeding rate and variety/hybrid relative maturity considerations for double crops compared to full-season.
| Crop | Seeding rate | Relative maturity |
| ???????? Difference between double crop and full-season ???????? | ||
| Soybean | Increase | No change or longer |
| Sorghum | Increase | Shorter |
| Sunflower | No change | Shorter |
| Corn | No change | Shorter |
Volunteer wheat control
One of the issues with double cropping that is often overlooked by producers is the potential for volunteer wheat in the crop following wheat. If volunteer wheat emerges and goes uncontrolled, it can cause serious problems for nearby wheat fields in the fall as a host for the wheat streak mosaic complex of viruses [wheat streak mosaic (WSMV), High Plains disease (HPD), and triticum mosaic (TriMV)] that are transmitted by the wheat curl mite (WCM).
Volunteer wheat can generally be controlled fairly well with glyphosate or Group 1 herbicides such as quizalofop (Assure II, others), clethodim (Select Max, others), or sethodydim (Poast Plus, others), but control is reduced during times of drought stress. Atrazine can provide control of volunteer wheat in double-crop corn or sorghum, but control can be erratic depending on rainfall patterns.
For more detailed information about herbicides, see the “2025 Chemical Weed Control for Field Crops, Pastures, and Noncropland” guide available online at https://www.bookstore.ksre.ksu.edu/pubs/CHEMWEEDGUIDE.pdf or check with your local K-State Research and Extension office for a paper copy. The use of trade names is for clarity to readers and does not imply endorsement of a particular product, nor does exclusion imply non-approval. Always consult the herbicide label for the most current use requirements.
To Subscribe to the KSU Agronomy E-Updates follow this link
https://eupdate.agronomy.ksu.edu/index_new_prep.php
Authors contributing to the post
Sarah Lancaster, Weed Management Specialist
slancaster@ksu.edu
John Holman, Cropping Systems Agronomist
jholman@ksu.edu
Logan Simon, Southwest Area Agronomist
lsimon@ksu.edu
Tina Sullivan, Northeast Area Agronomist
tsullivan@ksu.edu
Jeanne Falk Jones, Multi-County Agronomist
jfalkjones@ksu.edu
Sorghum Nitrogen Timing
Contributors:
Josh Lofton, Cropping Systems Specialist
Brian Arnall, Precision Nutrient Specialist
This blog will bring in a three recent sorghum projects which will tie directly into past work highlighted the blogs https://osunpk.com/2022/04/07/can-grain-sorghum-wait-on-nitrogen-one-more-year-of-data/ and https://osunpk.com/2022/04/08/in-season-n-application-methods-for-sorghum/
Sorghum N management can be challenging. This is especially true as growers evaluate the input cost and associated return on investment expected for every input. Recent work at Oklahoma State University has highlighted that N applications in grain sorghum can be delayed by up to 30 days following emergence without significant yield declines. While this information is highly valuable, trials can only be run on certain environmental conditions. Changes in these conditions could alter the results enough to impact the effect delay N could have on the crop. Therefore, evaluating the physiological and phenotypic response of these delayed applications, especially with varied other agronomic management would be warranted.
One of the biggest agronomic management sorghum growers face yearly is planting rate. Growers typically increase the seeding rate in systems where specific resources, especially water, will not limit yield. At the same time, dryland growers across Oklahoma often decrease seeding rates by a large margin if adverse conditions are expected. If seeding rates are lowered in these conditions and resources are plentiful, sorghum often will develop tillers to overcome lower populations. However, if N is delayed, there is a potential that not enough resources will be available to develop these tillers, which could decrease yields.
A recent set of trials, summarized below, shows that as N is delayed, the number of tillers significantly decreases over time. Furthermore, the plant cannot overcompensate for the lower number of productive heads with significantly greater head size or grain weight.
This information shows that delaying sorghum N applications can still be a viable strategy as growers evaluate their crop’s potential and possible returns. However, delayed N applications will often result in a lower number of tillers without compensating with increased primary head size or grain weight.



This date on yield components is really interesting when you then consider the grain yield data. The study, which is where the above yield component data came from, was looking at population by N timing. The Cropping Systems team planted 60K seeds per acre and hand thinned the stands down to 28 K (low) and 36K (high). The N was applied at planting, 21 days after emergence, and 42 days after emergence. The rate of N applied was 75 lbs N ac. It should be noted both locations were responsive to N fertilizer.

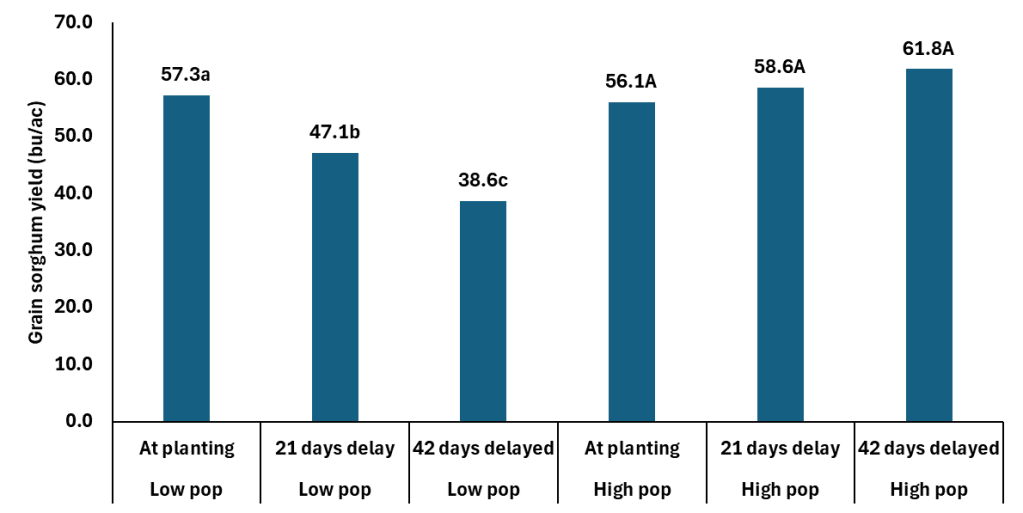
In the data you can without question see how the delayed N management is not a tool for any of members of the Low Pop Mafia. However those at what is closer to mid 30K+ there is no yield penalty and maybe a yield boost with delayed N. The extra yield is coming from the slightly heavier berries and getting more berries per head. Which is similar to what we are seeing in winter wheat. Delaying N in wheat is resulting in fewer tillers at harvest, but more berries per head with slightly heavier berries.
Now we can throw even more data into the pot from the Precision Nutrient Management Teams 2024 trials. The first trial below is a rate, time and source project where the primary source was urea applied in front of the planter for pre in range of rates from 0-180 in 30 lbs increments. Also applied pre was 90 lbs N as Super U. Then at 30 days after planted we applied 90 lbs N as urea, SuperU, UAN, and UAN + Anvol.
Pre-plant urea topped out at 150 lbs of Pre-plant (57 bushel), but it was statistically equal to 90 lbs N 51 bushel. The use of SuperU pre did not statistically increase yield but hit 56 bushel. The in-season shots of 90 lbs of UAN, statistically outperformed 90 pre and hit our highest yeilds of 63 and 62 bushel per acre. The dry sources in-season either equaled their in preplant counter parts.

The Burn Study at Perkins, showed that the N could be applied in-season through a range of methods, and still result good yields. In this study 90 lbs of N was used and applied in a range of methods. The treatments for this study was applied on a different day than the N source. Which you can see in this case the dry untreated urea did quite well when when applied over the top of sorghum. In this case we are able to get a rain in just two days. So we did get good tissue burn but quick incorporation with limited volatilization.

Take Home:
Unless working in low population scenarios. The data show that we should not be getting into any rush with sorghum and can wait until we know we have a good stand. We also have several options in terms of nitrogen sources and method of application.
Any questions or comments feel free to contact Dr. Lofton or myself
josh.lofton@okstate.edu
b.arnall@okstate.edu
Funding Provided by The Oklahoma Fertilizer Checkoff, The Oklahoma Sorghum Commission, and the National Sorghum Growers.