Home » Nitrogen (Page 2)
Category Archives: Nitrogen
Grain Sorghum, 2023 edition
As I sit in my office writing this I am seeing the chances for Stillwater to get a good rain today slowly dwindle away. Last night we had a 75% chance of 0.56. Its now 3pm we have received 0.01 and have a 60% chance of getting an additional 0.10. And as this is how 2023 has gone, but we are still better off than so many west and north of us.
No some of the zeroed out wheat ground and winter fallow ground has seen its first moisture in 100+ days. Planters and drilling are rolling and or ready to roll. So I wanted to throw out a few thoughts and re-share an older blog.
In August of last year I was talking about how the extended drought was impacting organic matter and nitrogen cycling “Nitrogen cycle hiccups and a lot of drying“. Wish I could say things have changed since then but we all now otherwise. While we have moisture to plant and germ our H2O fuel tank is far from full. Its going to take significant rainfall to rebuild the soil profile, not to mention the ponds. And I can not forget how last year we had great rains in May and were going pineapple by the end of July. I had N response studies all over the state in sorghum, at every trial nitrogen was not a limiting factor.
By this point if you know me or have every read anything I have written in the past you should know what is coming. My recommendation for pre-plant N is 0.0 lbs. Go ahead and put a starter down where you have low P or pH that’s out of range, and Fe and/or Zn in the calcareous soils where needed, but that’s it. We are continually adding to the research data base that says sorghum responds exceptionally well to in-season nitrogen applications in some cases nearly all the way to boot stage. I believe we are close to determining/explaining why the crop does so well but not ready to share that work just yet.
I just do not have the trust in the what rains may come to spend money on a fertilization pass that has been proven to be less valuable. Get the seed in the ground and crop up, put out some N-Rich strips. Let the weather play out for a month or so and see 1) Do I have soil moisture to get me through harvest or am I living day by day on a hope and a prayer? 2) is my N-Rich strip showing?
For Question 1) if your living day to day is further investment in the crop warranted? If you have soil moisture and rain in the forecast, its time to rock and roll. For question 2) which I hope you take my advice on doing. If the N-Rich is showing up 30 days into the you can use some rough yield goal estimations and apply 1 lb N per bushel or go get access to a GreenSeeker sensor (available through OSU extension offices) and use OSU’s online calculator SBNRC. If the strips not showing up then you need to consider that a month into the crops growth the soil has supplied 100% of the crops N need. Depending on if you want to push yield or save N adjust your rec accordingly. I would say reducing planned N rate by 40-50 lbs would a legitimate option. There are more and more agronomist utilizing N strips in wheat and sorghum so you could check around.
Final thoughts.
The more I do research on N timing and N management the more I am finding that there are some great benefits to limiting early N availability to the crop. To the point were I am finding and increasing value of an early season N stress on crop performance and grain yield.
Now for rehashed research. Much like wheat, sorghum can and probably should wait for N.
Can Grain Sorghum Wait on Nitrogen? One more year of data.
Original Posting 4.7.2022
Michaela Smith, Ph.D. candidate under advisement of B. Arnall
Brian Arnall, Precision Nutrient Management Specialist
The impressive ability of sorghum to recover from significant N stress with late applied nitrogen was originally reported in the blog “Can grain sorghum wait on nitrogen”. This projected was replicated again in 2021 and these are the results. During the 2021 growing season, frequency of rainfall events and amounts were similar to the 2020 growing season. Figure 1. shows the application dates and rainfall events.

Statistically there was no significant difference in grain yield from the pre-plant (0) and any application up to the 56 day application. This data matches up quite well the yield results from 2020.
Looking and the current soil moisture conditions (Figure 3.) and fertilizer price I think this data supports the recommendation to get the seed in the ground and see how the situation plays out. If there are decent rains at and shortly after planting then there is time to apply nitrogen with little risk of yield loss. If fact the weather during late May and early June provide some of the best chances to getting the fertilizer rained in (Figure 4) . If the weather doesn’t cooperate and provide us the much needed rains, then by waiting to apply we are not left with a lot of Expensive nitrogen setting out in the field of a failed crop.
Final Thoughts Heading into the 2021 Sorghum Planting Season.
– Make sure you have a soil sample, knowing P and K will be critical.
– No need to front load N fertilizer, especially if soil moisture is short.
– Utilize in-furrow P if soil pH and or soil test P is low.
– If you skip pre-plant N APPLY N-RICH STRIPS!!!!
– If you apply pre-plant N consider applying 50% of the expected N or less.
– If you are applying any level of pre-plant N, Create Zero-N Strips.
For more information or questions contact
Brian Arnall b.arnall@okstate.edu 405.744.1722
Special thanks to EDC Ag Products Co LLC for suppling NH4NO3 used in the delayed N project.
Original Post March 22, 2022.
Data for this post available in thesis Impact of delayed nitrogen application in grain sorghum
Smith, Michaela Lynn (2021-05). Available at https://shareok.org/discover
Grain sorghum producers in Oklahoma are challenged greatly by their environment and sporadic rainfall patterns, which diminish as the season progresses. These uncontrollable variables influence timing of nitrogen (N) application and nitrogen use efficiency. Using rainfall events as an incorporation method forces producers to apply before the event regardless of its intensity or delay application until field conditions are acceptable while anxiously waiting for another rainfall event. When deciding to delay N application it’s important to know the effects on physiological development and grain yield.

Trial structure and breakdown
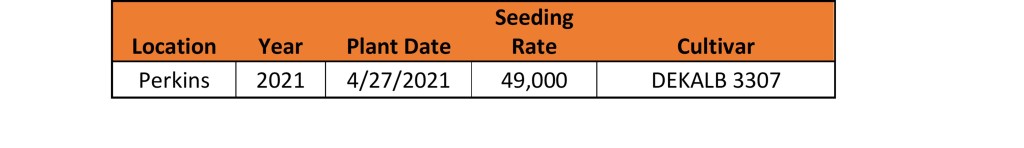
This study was conducted over the 2020 growing season consisting four locations, including one double cropping system following wheat. Ten in-season applications were made using ammonium nitrate (AN) as the N source at a rate of 90 lbs. ac. Using AN as the N source reduced the risk of nitrogen loss through the process of volatilization as the goal of the research was to test the plant not the fertilizer. A pre-plant treatment served as the standard check, while in-season applications were initiated at 21 Days After Planting (DAP) and applications made sequentially at 7-day intervals. A non-fertilized check was included to the study to confirm locations were responsive to N fertilized applications Hybrid, plant date, and seeding rate can be found in Table 1.

Physiological Response to Application Timing
Two of the four locations demonstrated an effect to physiological development and maturity with the delay of nitrogen application. A delay in heading by a one to two-week period was observed at Perkins and Lahoma for applications made after May 21st (Table 2.). This delay in heading contributed to similar delay in maturity and potential harvest date. At Perkins decreased plant height was observed in the pre-plant plot and was associated with the onset of late season nitrogen deficiency (Figure 2). While this response was unexpected, the impact of nitrogen deficiency experienced early in the crop growth on the root and shoot growth has been well documented in many species. As a plant experiences nitrogen limitations growth changes from above ground to the below ground parts (roots) in an attempt to alleviate nitrogen stress. This increase in root growth could contribute to a more efficient uptake of nitrogen and decrease loss. In contrast to Figure 2, pre-plant application is shorter than compared to later season applications, this could be a result of inadequate N uptake thus leading to N loss by leaching, whereas later applications had increased root growth for efficient N interception and uptake.


Yield Response to Application Timing
Response of N was observed at all locations (Figure 3), while the delay of nitrogen varied in its effects across all locations. Grain yield from each N application was compared back to the pre-plant application to evaluate the effects of timing. All four locations responded positively to N fertilizer. At both LCB and Lahoma grain yield was maintained with applications made as late as 42 to 63 DAP respectively before any negative trend in grain yield was observed. Perkins was the only locations to have a statistically significant increase in grain yield due to delayed N applications. At this site, which is a sandy loam, waiting until 42 DAP resulted in a 15 bushel increase over the pre-plant plot. Now Alva which was double crop showed that rainfall is key. At this site, none of the in-season treatments made it up the level of the pre-plant. The reason for this will be discussed further below.

Influence of Rainfall
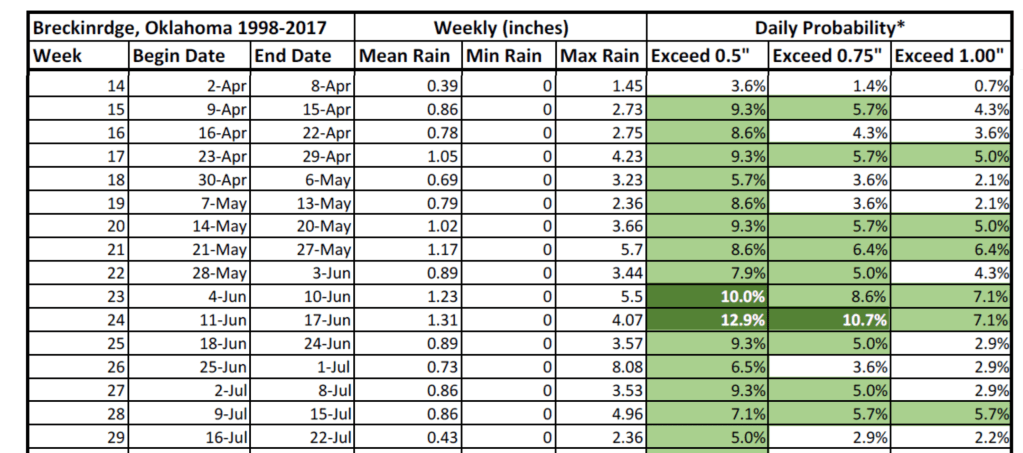
The loss in grain yield at Perkins in the pre-plant application could likely be reflective of nitrogen loss due to leaching. Pre-plant applications have been well documented in the aspect loss as a result of crop requirement and early physiological development. Long term mesonet rainfall data depicts a decline in the probability of rainfall with the progression of the growing season across all locations. In early season the probability of 0.5 inches of rainfall ranges from 8 to 10% respectively for LCB, Lahoma, and Perkins, and dramatically decline to percentages at low as 5% in mid-July during grain filling period. For Alva rainfall probability is substantially lower as its season was initiated during the drier months, which depicted a probability of 6% for 0.5 inches of rainfall, and 4.5% for 1 inch for early season rainfall crucial for pre-plant incorporation and crop establishment. These probabilities drop considerably compared to regular season as the months progress onward, mid to late August probability for 0.5 inches ranges from 0.8 to 11.5%, while for a 1 inch is 0 to 6.9%. Past weather data provided by the mesonet illustrates how later in the season rainfall and its amount is variable, suggesting that in a double crop scenario delayed application is not recommended while it is in regular season crop due to the increased chance of rainfall probability.
Summary
The purpose of this study was to evaluate the impacts of delayed nitrogen application in grain sorghum. In order to develop an accurate conclusion additional site years are required, although current data could suggest delaying nitrogen application for full season grain sorghum is possible without a detrimental loss in grain yield. This means producers have time to evaluate the crop and market to determine if more inputs are needed and economical, while allowing implementation of technologies such as the N-Rich Strip and SBNRC.
If you have any questions for comments please reach out.
Brian Arnall
b.arnall@okstate.edu
405.744.1722
Acknowledgement of EDC Ag Products Co LLC for support of this project.
In-season N application methods for Sorghum
Raedan Sharry, Ph.D. candidate under advisement of B. Arnall
Brian Arnall, Precision Nutrient Management Specialist
The data about to be reported is from the study we have fondly named “Burn Baby Burn”, you will see why soon enough.
Grain Sorghum production continues to be an important component of many growers crop rotations in the Great Plains. However, for many growers who focus primarily on small grains production, equipment restraints may impose limits on in season nitrogen (N) management. When producers are able to delay the application until in-season it helps to ensure that N is available to the crop at the time of increased uptake during the reproductive stages of the crops life. Producers often have access to equipment and technologies that may be used to take advantage of improved N application timing, but may worry about the negative effects that nitrogen can have if the fertilizer is inadvertently applied to plant material. An experiment was initiated in Central Oklahoma to evaluate the yield response of grain sorghum to in-season nitrogen application methods.
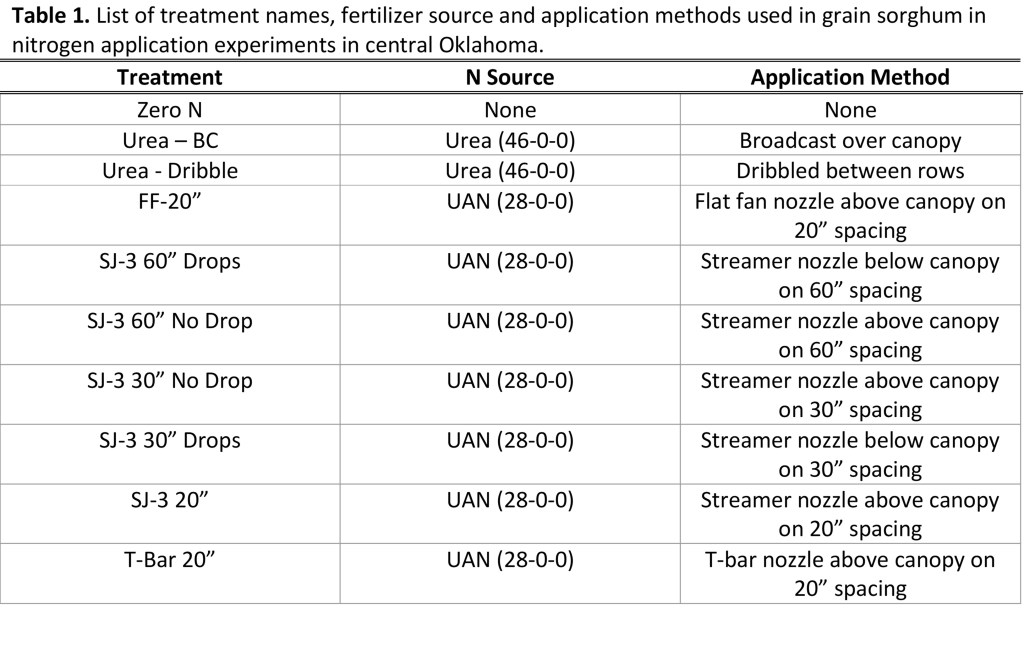
Trials were placed at Lake Carl Blackwell near Stillwater, Perkins and Chickasha Oklahoma and included 9 in-season fertilization methods and a 0 nitrogen control. Treatments are listed in Table 1 below.
In total 120 lbs of N was applied to all treatments receiving in-season applications. 60 lbs was applied at planting to all treatments including the “Zero N Control”. The remaining 60 lbs. of N was applied according to application method in-season. The urea was applied by hand and the liquid treatments a push cart with adjustable boom height (Figure 1) was used to apply the UAN. Applications were made mid day at V8 growth stage. The temperature at the time of all applications was about 90 F and humidity below 75%. Nozzle position for 30″ and 60″ was set for between rows.
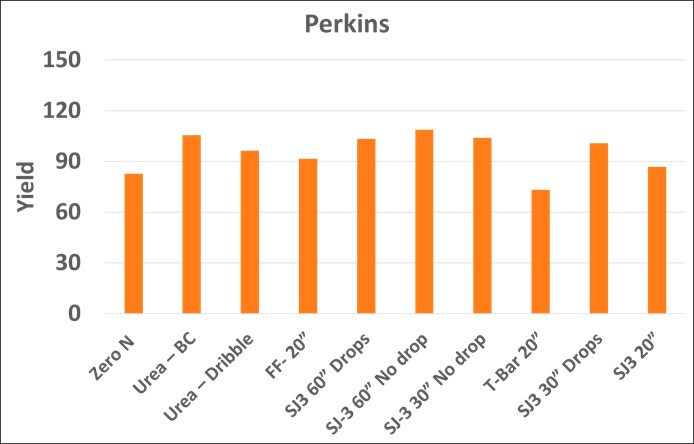
At two of the three locations (Stillwater and Perkins) the addition of 60 lbs. of N in-season increased yield above the control treatment. At the Stillwater (Lake Carl Blackwell) location there were no statistical differences (α=0.05) between in-season fertilized treatments except the T-Bar 20” treatment (Figure 2). The Perkins location (Figure 3) provided a similar result in which again there was no statistical difference between fertilized treatments, excluding the T-Bar 20” treatment.
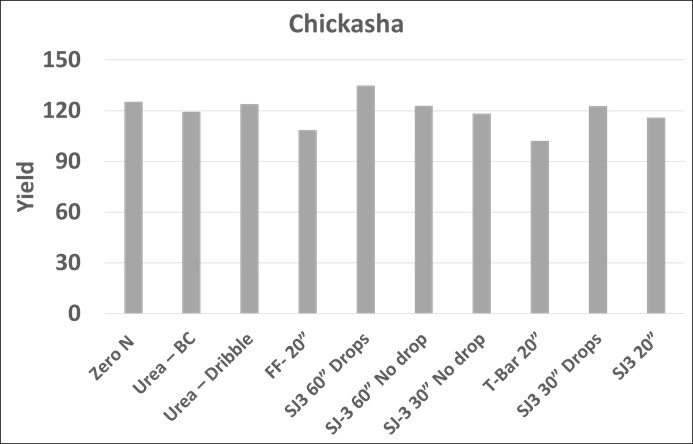
The Chickasha location differed in that additional in-season nitrogen did not improve yield (Figure 4). While we want a response to applied N, in the case it allows use to solely evaluate the impact of burn associated with N application. The T-bar 20” treatment statistically negatively impacted grain yield and the FlatFan-20″ did at α=0.10, which means we are only 90% confident the yield lose was due to treatment. This response has been consistent across all three locations, on average decreasing yield approximately 21 bu/ac relative to the individual site grain yield average.
Even though it was mentioned for Chickasha, it is also important to note that while it was not statistically significant (α=0.05) the FF- 20” treatment (Flat Fan nozzles above canopy on 20” spacing) trended towards decreasing yields at all 3 locations and is likely detrimental to crop performance. At all locations substantial damage to leaf material was observed, similar to that pictured in Figure 5 below. Several of the treatments damaged leaf material on the plant through burn injury, but most were not negatively impactful on grain yield in the 2021 growing season. Grain sorghum yield did not benefit from moving the application point below the canopy using drop attachments, nor did adjusting nozzle spacing from 30 to 60”. Source was not a significant factor impacting grain yield regardless of it application method.

The observations from this study show that many of the in-season nitrogen application methods that are available to growers will not negatively impact yield. This however does not apply to tools such as the T-Bar. Similar tools that concentrate large amounts of N to leaf material are also likely to produce similar results. It is important to note that the T-bar was used on 20” spacings and not tested otherwise. Moving the spacing of the T-bar may lead to different results.
Growers who are looking to move N applications in their grain sorghum crop to in-season to capture the benefits associated will likely be able to with equipment that is already available to them. While leaf damage may be observed under sub-optimal application methods, damage is unlikely to contribute to significant yield loss. However, growers should keep in mind that environmental conditions may have a significant impact on the results seen from these types of application as growers should always look to limit stress to the plant when possible.
We of course will be putting out a second year of this study and will share the results when we can.
For more information or questions contact
Brian Arnall b.arnall@okstate.edu 405.744.1722
Can Grain Sorghum Wait on Nitrogen? One more year of data.
Michaela Smith, Ph.D. candidate under advisement of B. Arnall
Brian Arnall, Precision Nutrient Management Specialist
The impressive ability of sorghum to recover from significant N stress with late applied nitrogen was originally reported in the blog “Can grain sorghum wait on nitrogen”. This projected was replicated again in 2021 and these are the results. During the 2021 growing season, frequency of rainfall events and amounts were similar to the 2020 growing season. Figure 1. shows the application dates and rainfall events.


Statistically there was no significant difference in grain yield from the pre-plant (0) and any application up to the 56 day application. This data matches up quite well the yield results from 2020.
Looking and the current soil moisture conditions (Figure 3.) and fertilizer price I think this data supports the recommendation to get the seed in the ground and see how the situation plays out. If there are decent rains at and shortly after planting then there is time to apply nitrogen with little risk of yield loss. If fact the weather during late May and early June provide some of the best chances to getting the fertilizer rained in (Figure 4) . If the weather doesn’t cooperate and provide us the much needed rains, then by waiting to apply we are not left with a lot of Expensive nitrogen setting out in the field of a failed crop.
Final Thoughts Heading into the 2021 Sorghum Planting Season.
– Make sure you have a soil sample, knowing P and K will be critical.
– No need to front load N fertilizer, especially if soil moisture is short.
– Utilize in-furrow P if soil pH and or soil test P is low.
– If you skip pre-plant N APPLY N-RICH STRIPS!!!!
– If you apply pre-plant N consider applying 50% of the expected N or less.
– If you are applying any level of pre-plant N, Create Zero-N Strips.

For more information or questions contact
Brian Arnall b.arnall@okstate.edu 405.744.1722
Special thanks to EDC Ag Products Co LLC for suppling NH4NO3 used in the delayed N project.
Original Post March 22, 2022.
Data for this post available in thesis Impact of delayed nitrogen application in grain sorghum
Smith, Michaela Lynn (2021-05). Available at https://shareok.org/discover
Grain sorghum producers in Oklahoma are challenged greatly by their environment and sporadic rainfall patterns, which diminish as the season progresses. These uncontrollable variables influence timing of nitrogen (N) application and nitrogen use efficiency. Using rainfall events as an incorporation method forces producers to apply before the event regardless of its intensity or delay application until field conditions are acceptable while anxiously waiting for another rainfall event. When deciding to delay N application it’s important to know the effects on physiological development and grain yield.

Trial structure and breakdown
This study was conducted over the 2020 growing season consisting four locations, including one double cropping system following wheat. Ten in-season applications were made using ammonium nitrate (AN) as the N source at a rate of 90 lbs. ac. Using AN as the N source reduced the risk of nitrogen loss through the process of volatilization as the goal of the research was to test the plant not the fertilizer. A pre-plant treatment served as the standard check, while in-season applications were initiated at 21 Days After Planting (DAP) and applications made sequentially at 7-day intervals. A non-fertilized check was included to the study to confirm locations were responsive to N fertilized applications Hybrid, plant date, and seeding rate can be found in Table 1.

Physiological Response to Application Timing
Two of the four locations demonstrated an effect to physiological development and maturity with the delay of nitrogen application. A delay in heading by a one to two-week period was observed at Perkins and Lahoma for applications made after May 21st (Table 2.). This delay in heading contributed to similar delay in maturity and potential harvest date. At Perkins decreased plant height was observed in the pre-plant plot and was associated with the onset of late season nitrogen deficiency (Figure 2). While this response was unexpected, the impact of nitrogen deficiency experienced early in the crop growth on the root and shoot growth has been well documented in many species. As a plant experiences nitrogen limitations growth changes from above ground to the below ground parts (roots) in an attempt to alleviate nitrogen stress. This increase in root growth could contribute to a more efficient uptake of nitrogen and decrease loss. In contrast to Figure 2, pre-plant application is shorter than compared to later season applications, this could be a result of inadequate N uptake thus leading to N loss by leaching, whereas later applications had increased root growth for efficient N interception and uptake.


Yield Response to Application Timing
Response of N was observed at all locations (Figure 3), while the delay of nitrogen varied in its effects across all locations. Grain yield from each N application was compared back to the pre-plant application to evaluate the effects of timing. All four locations responded positively to N fertilizer. At both LCB and Lahoma grain yield was maintained with applications made as late as 42 to 63 DAP respectively before any negative trend in grain yield was observed. Perkins was the only locations to have a statistically significant increase in grain yield due to delayed N applications. At this site, which is a sandy loam, waiting until 42 DAP resulted in a 15 bushel increase over the pre-plant plot. Now Alva which was double crop showed that rainfall is key. At this site, none of the in-season treatments made it up the level of the pre-plant. The reason for this will be discussed further below.

Influence of Rainfall
The loss in grain yield at Perkins in the pre-plant application could likely be reflective of nitrogen loss due to leaching. Pre-plant applications have been well documented in the aspect loss as a result of crop requirement and early physiological development. Long term mesonet rainfall data depicts a decline in the probability of rainfall with the progression of the growing season across all locations. In early season the probability of 0.5 inches of rainfall ranges from 8 to 10% respectively for LCB, Lahoma, and Perkins, and dramatically decline to percentages at low as 5% in mid-July during grain filling period. For Alva rainfall probability is substantially lower as its season was initiated during the drier months, which depicted a probability of 6% for 0.5 inches of rainfall, and 4.5% for 1 inch for early season rainfall crucial for pre-plant incorporation and crop establishment. These probabilities drop considerably compared to regular season as the months progress onward, mid to late August probability for 0.5 inches ranges from 0.8 to 11.5%, while for a 1 inch is 0 to 6.9%. Past weather data provided by the mesonet illustrates how later in the season rainfall and its amount is variable, suggesting that in a double crop scenario delayed application is not recommended while it is in regular season crop due to the increased chance of rainfall probability.
Summary
The purpose of this study was to evaluate the impacts of delayed nitrogen application in grain sorghum. In order to develop an accurate conclusion additional site years are required, although current data could suggest delaying nitrogen application for full season grain sorghum is possible without a detrimental loss in grain yield. This means producers have time to evaluate the crop and market to determine if more inputs are needed and economical, while allowing implementation of technologies such as the N-Rich Strip and SBNRC.
If you have any questions for comments please reach out.
Brian Arnall
b.arnall@okstate.edu
405.744.1722
Acknowledgement of EDC Ag Products Co LLC for support of this project.
Utilizing N fixing biologicals.
In the past couple years significant efforts have been made to produce N fixing microorganisms that can be utilized in an agriculture system. The atmosphere is 78% N2 and prokaryotic microorganisms such as the bacteria species Azotobacter, Bacillus, Clostridium, and Klebsiella take that N2 gas and turn it into plant available NH4. These organisms have been around providing nitrogen for plants, for as long as there has been plants. In agriculture we have heavily utilized their relationship with legumes however have struggled bringing them into other realms of production. Naturally they tend to be found in areas that are very low levels of nitrogen. For example, prokaryotes were found in the un-fertilized check of the 130-year-old Magruder Plots but are not found any other treatment that receives fertilizer organic or commercial.

Now there are several products marketed as containing N fixing microorganisms suited for use in today’s corn, sorghum, and wheat production. While I have an active research program evaluating the use of such materials in Oklahoma, this blog will not address what works or how well. This blog will touch upon my thoughts on how to utilize a technology such as this if you pull the trigger to implement.
So there is one key to getting a ROI on products that create plant available nitrogen, and it’s a really simple key.
Under Apply Nitrogen
If you apply enough or more N than the crops needs, then there is ZERO value in a product that creates more N. For example, applying one of these products in your 250-bushel yield goal corn after you’ve already laid down 300 lbs of N preplant. Unless you lose it all to leaching, your probability of seeing a ROI on your biological investment is pretty poor. I have a hard time understanding the thought process behind paying for a N fixing product and not lowering your fertilizer rate. I can see one of two reasons. 1) You believe you historically under apply N and are losing yield because of such 2) Are in an environment which has a high potential of late season N losses, and you are unable to make recovery applications.
So what to do if using a N Fixer? I do not have the confidence yet to say, “Apply X product, it will produce Y lbs of N, so cut your rate by Y lbs”. That uncertainty is one of the greatest challenges, not knowing will I get 10 lbs or 40 lbs? If I did, then I would just subtract that off my planned rate. Side note, as someone who has been doing on farm N rate studies for a decade plus, I would have to add that most were likely over applying by that much and could cut back anyways. For me the use of the N Fixers should force your hand into utilizing in-season N applications, regardless the crop. So that you can better predict or determine impact of the product.
This is where the use of a refence strip (N-Rich or Zero N) is the golden ticket. We need a way to quickly evaluate the amount of N the crop has access to. The N-Rich method works best when preplant N is drawn way back. I would add that reduced pre-plant is a great scenario for N Fixers. The N-Rich in comparison to the rest of the field will provide you guidance towards your in-season goals. If the N-Fixers are doing a great job the N-Rich will not be showing up any time soon and you can make your N rate adjustments accordingly. If you are a Pre-plant or die kind of farmer, then I say you need to pull back the reins on the preplant rate but give the N Fixers some room to add value and add in your Zero N strips. These will again let you observe what is happening in the soil apart from your fertilizer. If it is getting on the late side of in-season N and you cannot find your zero, might be a good time to walk away and hang up the fertilizer applicator keys. I have lots of blogs and pubs on the use of reference strip so send me a note if you want to dive further into these approaches.

Feel free to reach out with questions or comments. B.arnall@okstate.edu
Related Blogs
Its dry and nitrogen cost a lot, what now?
The title says a lot about the primary question I am receiving right now. And the latest long range “forecast” does not make me feel any better about the current situation. But it is what it is and many great plains wheat farmers are having to make a decision.
The current situation in the wheat belt is that we are dry to depth, when the 32 inch PAW is on short supply and this comes from a combination of no rain and above average temperatures.

Fertilizer prices are holding fairly strong, at expensive, and the wheat crop currently seems to be going in reverse. So what is a wheat farmer to do? If we are looking on the bright side the lack of moisture in the surface will help reducing any potential losses through urea volatilization. It does not make the potential for loss zero though. If I am bound and determined to fertilize now, I would be very selective of the source and method of application. The biggest driver, tillage and residue amounts.
- Conventional Till / No residue (plenty of bare soil showing) and small wheat-
- UAN via Streamer nozzles
- Why: With UAN (urea ammonium nitrate) you have a liquid N source that will get onto and into the soil and readily available nitrate. Streaming on will help concentrate the fertilizer and potential reduce any urea volatilization if any dews were to occur. Urea would sit until dissolved and lead to potential losses if the first moisture was heavy dew and not a incorporating rainfall.
- UAN via Streamer nozzles
- No-till / high residue (no bare soil showing)-
- Dry Urea
- Why: If Our residue is dry when the urea is spread the wind will help push it below the residue surface providing protection until a good rain. If UAN is applied to this dry or even slightly damp residue and not washed off with a rainfall in a week or so the amount of N tied up in that residue will likely be significant.
- Dry Urea
- The big wheat (very little bare soil, lots of wheat tissue.
- Urea or UAN Streamer
- Why not Flat fan. At least with the current status the wheat is not growing and bigger wheat has increasing levels of tip die back. So while UAN sprayed on actively growing wheat can be absorbed foliarly, stressed wheat can not do it as well. Plus the UAN that hits dead or damaged tissue will not make it into the plant. The UAN applied via flat fan will need incorporation via rain in a couple days.
- Urea or UAN Streamer
You may have caught in the paragraph above I said, “If I was bound and determined”. If I had the option I am not pulling the trigger until after I have received some good moisture. I fully expect and have already seen rigs running before every decent chance of rain. Unfortunately many of those chances have not panned out and that will remain my concern moving forward. I want to make sure we have some water in the tank before investing in the system.
But now we increase the risk/fear by waiting and the question I get is what if we don’t get good rains or don’t get good incorporating rains. The short answer is, if we don’t get rains the N application is the least of our concerns. If we approach March 15th and we have not had the rains needed to put a little water in the tank and incorporate the N then we are not likely looking at a bumper crop which will need N. What survives in that scenario will be living off deep soil water, and where there is deep soil water there is a good chance of deep N. The shallow soils will be so stressed that nutrient demand will be very little.
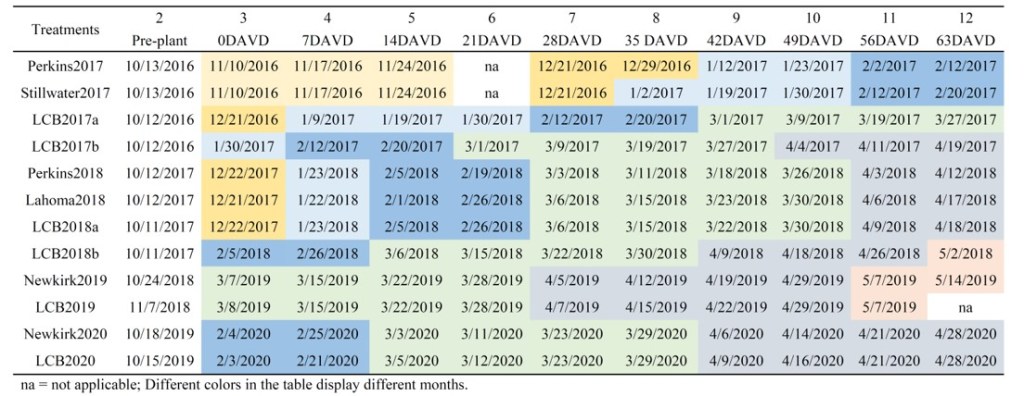
Now lets talk waiting and applying N. How late before we just say we are done. To answer I am going to draw from a data set I talk about a lot, the delayed N work by Dr. Souza. This study was started in the fall of 2016 and concluded with the 2020 wheat harvest. In all, twelve trials were established and achieved maturity. This study was designed to evaluate the recovery of winter wheat grain yield and protein after the crop was N stressed. Treatments included an untreated check, pre-plant application and ten in-season treatments. The application of in-season treatments was initiated when N deficiency was confirmed and treatments were applied in progressive order every seven growing days to the point of 63 growing days after visual deficiency (DAVD). A growing degree days is any day that the average daily temperature is at or above 40⁰ F. Ammonium nitrate (NH4NO3) was applied at a rate of 90 lbs N ac-1 for all treatments.
With this data we can answer two questions, first at what point did we lose yield compared to pre-plant and second how late could we apply and still increase yield above the check. So comparing to the pre lets us know how long could we wait with losing yield. Across the trials we lost yield three times by waiting too long, at LCB2017b that was 4/19, Lahoma18 it was around 3/30, and then Newkirk2020 we lost yield by waiting until 4/6. This data is why I am pretty comfortable waiting until mid March when and if needed. Now if we look at the check, that will tell us if things start improving late can we get still get a yield bump with added N. Newkirk 2020 was the only time and place we could increase yield above the zero after the 4/14 additions.

Take Home Message
My recommendation is that if you are not required to take delivery or needing to cover a lot of acres, i.e. time limited, I would not get in a hurry to apply N on this wheat crop. I think if we combine weather by market this a good time to wait and see. Once we get a rain and have some soil moisture it will be time to run the rigs. The crop currently does not need a lot of N so why spend the $. If things don’t improve by mid to late march, consider the wheat a cover and look towards a summer crop with the hopes of rains in April. If you need to take the crop to yield, then you can wait a while longer and still get a return on the N, with hopes the price could come down a bit.
Finally, While I don’t suggest running fertilizer in front of the first chance of rain, I would make sure I had an N-Rich strip on each and every single field. Strips can go out well past green up and serve a great purpose. The N-Rich strip will help you determine if the crop is able to mine any soil N or if the N tank is dry.
Feel free to reach out with questions or comments.
Brian Arnall Precision Nutrient Management Specialist.
b.arnall@okstate.edu
Special thanks to EDC Ag Products Co LLC for suppling NH4NO3 used in the delayed N project.
Relevant past blogs for your reading enjoyment.
The Easy Button for Nitrogen…….
Brian Arnall, Precision Nutrient Management Extension Specialist.
The basics for nitrogen (N) fertilizer rate determination can be described in a mechanistic approach by the Stanford Equation NFert = ( NCrop – NSoil ) / Neff. This equations states that the N fertilizer rate is equal to the amount of nitrogen taken up by the crop minus the amount of nitrogen supply by the soil, divided by the efficiency of the nitrogen fertilizer used. I outline the importance of this equation in the blog “Components of a variable rate nitrogen recommendations“.
There are nitrogen “Easy Buttons” which utilizes averages collected over diverse environments to create accurate N rate recommendations. The best example of this is the yield goal rules of thumb such as wheats 2.0 lbs N per yield goal bushel minus soil test nitrate. Yield goals are generally calculated as the average of the best 3 out of 5 years, or the 5-year average times 20%. Also, the 2.0 lbs of N is more than what is in a bushel as it also adds in an efficiency factor or a 0.5 lbs per bushel cushion. This method and others like it provide an accurate N rate with slight probability of yield loss. However, the rec is often highly imprecise. Meaning that if I apply the method to 100 fields the average will be spot on, however if I look at the performance of the recommendation on a single field, I will likely be disappointed.

When it comes to nitrogen recommendations the Easy button method will use components which help ensure that the rate prescribed will maximize yield 90-95% of the time. For example, take the data presented in Figure 2. Over fifteen years of the long-term winter wheat fertility study near Lahoma, Oklahoma the average pounds of N per bushel to reach economic optimum nitrogen rate (EONR) was 1.6, however if 2.0 of N was applied per bushel yield would have been maximized 13 out of the 15 years. While 2.0 lbs. of N per bushel would have been quite accurate for maximizing yield, it would be highly imprecise as over the 15 years optimum pounds of N per bushel ranged from 0.0 to 3.2.

The trick to improving your N rate recommendation closer to a precise and accurate system is to obtain representative site-specific values for the Stanford Equation NFert = (NCrop – NSoil) / Neff.
Looking at the 15-year long-term data above the yields range from a low of 27 to a high of 88 bushels. Of those 15 years, I personally planted multiple years, usually sometime in October, and many of those years while sowing I could have guessed a range of 55-60 bushel, which just happened to be just above the 15-year average. It was not until February and March when the yield potential really started to express itself. Why, well there is a lot of weather between Oct to March, a lot of environmental positive and negative impacts on that final grain yield. This is the best timing to go out with approaches, models, or techniques to estimate yield potential for N rate recs.
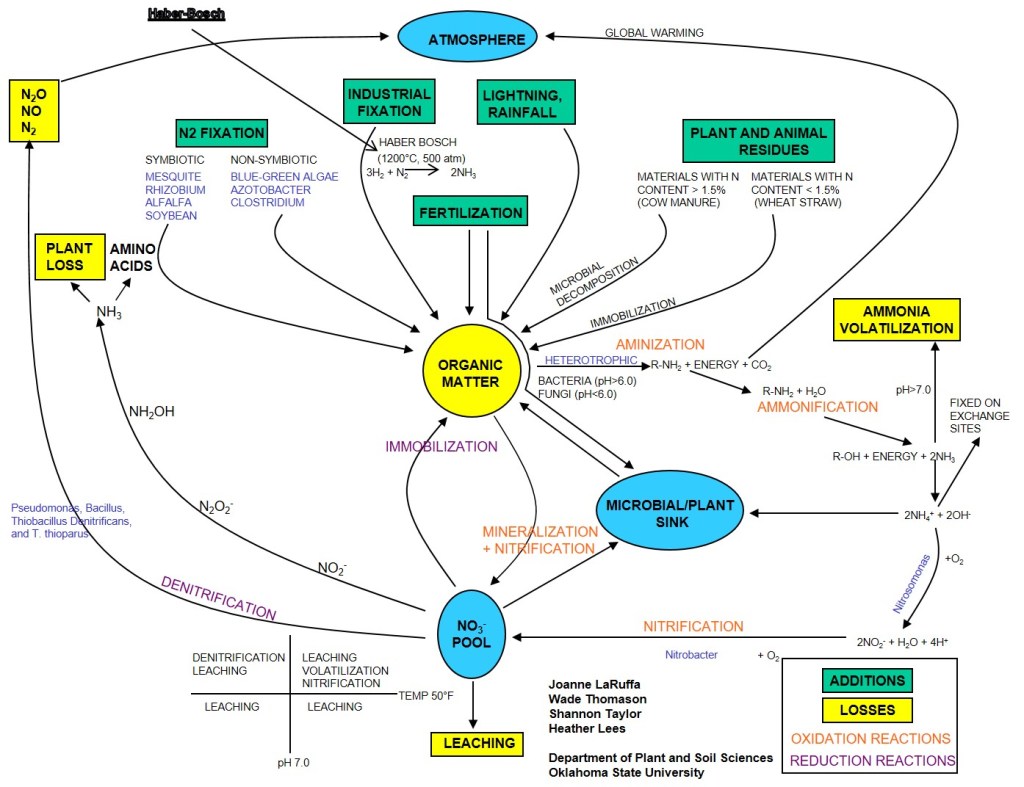
While I am a big fan of soil testing, pre-plant soil samples for N are just a snap shot in time. But the While I am a big fan of soil testing, pre-plant soil samples for N are just a snapshot in time, but the nitrogen cycle Figure 3, will roar on after the soil sample is collected. Organic matter (OM) is the central component of this cycle and drives availability of NH4 and NO3 in the system. For each 1% OM in the top 6″ of the soil there is approximately 1000 lbs of organically bound N. The amount of N going into and out of OM pool is driven by C:N ratio of residues, soil temperature and soil moisture. While we very well what the mechanisms of the cycle are and can model the reactions quite well. Our inability to predict long term weather patterns is the greatest factor limiting our ability to predict future availability of NSoil.
This is where the reader should be asking “how can we get better site specific data” and I begin the discussion on why I have been promoting the of the Sensor Based Nitrogen Rate Calculator (SBNRC) and N-Rich strip method.
Lets talk about how the approach follows Stanford’s mechanistic approach to N management. First the Yield Potential component of the SBNRC which is related to NCrop. In effect researchers have built models over the past two decades that can correlate the NDVI collected from a sensor, such as the GreenSeeker, with the crops biomass and chlorophyll content. If given the number of days the crop has been growing it is possible to use the NDVI collected from the crop as a tool to predict final grain yield. The closer the wheat gets to hollow stem, or the corn gets to tassel, the better the prediction. One reason is that we have allowed more “environmental influence” to happen. Dr. Bill Raun, a founder of the SBNRC concept kept great discussion and data sets on his NUE.OKSTATE.edu website. On the “NUE Website on YP” he provides information on how yield prediction work while on the “NUE Website YP Library” he has not listed every algorithm created, and the math behind them, but also a recipe book for how anyone can create their own algorithm. While there are a lot post sensing stresses that can bring down final grain yield, the models that have been built and continually improved, do quite a good job on predicting final grain yield in-season. Resulting a much more site specific value for NCrop. The blog”Sensing the N-Rich Strip and Using the SBNRC” goes into a further discussion of using the online SBNRC.
That now leaves NSoil, which I will argue is at least as important as NCrop. As weather so greatly influences the nitrogen cycle it would be nice to have a weather station on every field paired with a 0-4 ft soil description which could be incorporated into a model. Given those might be out of reach we have found the the use of a reference strip, high N or low N, really provides an site specific estimate the of nitrogen the crop has access to. If the high N reference (N-Rich) strip is showing up that means the remainder of the field is N deficient. This may be due to losses or lack of mineralization, either way more N is needed. If the N-Rich strip is not evident then the crop is finding enough N outside of the reference strip to support its current growth. This could be that residual N or mineralization is high, or it could mean that crop growth and therefore N demand is low. Having the N check strip in each field allows for a season long evaluation. We can use NDVI to characterize how big or little of a response we have to N. We call this the Response Index (RI). An RI of 1.8 means that we could increase yield by 80% if we add adequate N, if the RI is 1.05 then we are looking at a potential increase of 5%. I have a previous blog which goes into the application of the reference strip. “Nitrogen Rich Strips, a Reminder“
Finally we combine the two, YP and RI. By predicting the yield of the area out side the N-Rich strip we can determine environmental yield potential, YP0. Basically what can the field yield if nothing is added. We multiple YP0 by the RI to get the yield potential with added N, YPN. Then its as simple as N rate = (YPN – YP0 ) x N needed per bushel. So for example if YP0 is 40 bushel RI =2, then YPN is 80 bushel. I need to fertilize the additional 40 bushels of wheat and I can use the 2.0 N per bushel can come up with a top-dress rate of 80 lbs N per acre. We are now incorporating site specific in-season NCrop and NSoil data.
And just a reminder for those of you new to my blog, I have a lot of research documenting that it is not only OK, but often best if we wait on N application in wheat and other crops. Value of In-Season N blog.
Every step we take towards the easy button is often a step towards site specific imprecision due to the use of generalized terms or models. Depending on your goals this very well could be acceptable for your operation, but with nitrogen prices as volatile as they are, should we not be considering pushing the easy button to the side, for now. Let’s add a bit of site-specific data so that we can take advantage of the N the system may be giving us, or the yield we did not expect. Let the N-Rich Strip be that first step.
Relevant Peer Review Publications.
If you have any questions please feel free to contact me @ b.arnall@okstate.edu
Nitrogen Source: What’s “cheap” now may be lost later
Raedan Sharry, Ph.D. Student, Precision Nutrient Management
Brian Arnall, Extension Specialist, Precision Nutrient Management
Note, this blog is focused on grain only winter wheat production.
Crop producers looking to increase profits often consider how to reduce costs without sacrificing yield and/or quality. This applies to essentially all production functions including nitrogen application. Winter wheat growers in the southern Great Plains have a wide number of options available to them when considering nitrogen source and application technique. At the time of writing (08/27/2021) fertilizer prices obtained from the Two Rivers Farmers Cooperative are as follows ($/unit): UAN (28-0-0) $0.62, NH3 (82-0-0) $0.45, and Urea (46-0-0) $0.62. These price levels equate to approximately a 57% increase in urea cost, 65% increase in UAN28, and a 65% increase in NH
Application Timing
Winter wheat producers in the southern plains have historically applied nitrogen (N) fertilizer prior to planting, often utilizing anhydrous ammonia for application due to its generally lower price point per unit of N relative to other sources. However, research at Oklahoma State shows that if the total N application is delayed until approximately feekes 5 to feekes 7 stages (jointing) yields were increased 23% of the time while grain protein was increased 68% of the time. By delaying N application to later in the growing season N is more likely to be available when the crop requires by avoiding conditions conducive to losses. Further reading on delaying nitrogen application can be found here (https://osunpk.com/2020/09/10/value-of-in-season-application-for-grain-only-wheat-production/)
A study located a Perkins, OK observing yield and protein response provides an example of an expected response to delayed N. In this study 3 N fertilizer rates (180, 90 and 45/45 split) across 5 different timings (Pre, 30, 60, 90, and 120 days after planting) where investigated. Grain yield was maximized by the 180 lb. rate applied 60 days after planting, while protein was maximized at the 120 days after planting timing. This same trend continues across all N rate levels as the later N applications whether at 60 or 90 increased yield relative to the pre while the 120 days after planting application maximized protein level regardless of rate level. However, maturity of the 120 day application treatment was severely delayed. This experiment shows the ability to sustain yield while decreasing N rate if N application is pushed to later in the season to avoid conditions that lead to N losses as displayed by the 90 lbs. at 90 days after planting treatment compared to the 180 lb. pre-plant rate.

Application Cost
Application costs are directly related to choice of source utilized. For instance; anhydrous ammonia application is predicated on the use of a pulled implement such as a low disturbance applicator for in-season application or a tillage implement for pre-season application. This is compared to other sources such as urea or ammonium nitrate which may be broadcast, or UAN that can be applied using a sprayer. The relationship between source and cost of application is inherently related to the application efficiency of the equipment used. Table 2 below provides a rough idea of cost associated with different application methods. (Information Retrieved from Iowa State). Fuel cost assumed at $2.60/gal. Labor cost assumed to be $15.00/hr.
| Implement | Operating Efficiency | Fuel cost/ac | Labor Cost/ac | Operating cost/ac |
| 90’ SP Sprayer | ~78 ac/hr | $0.34 | $0.19 | $0.53 |
| 60’ Dry Spreader | ~30 ac/hr | $0.39 | $0.50 | $0.89 |
| 35’ Sweep Plow | ~21 ac/hr | $1.43 | $0.71 | $2.14 |
In many operations across the southern plains efficiency has become a key factor in decisions such as input selection and equipment purchases. This has come in response to the need to cover more acres with less labor. With that in mind and looking back to table 2 it is easy to see that a self-propelled sprayer is likely able to cover more acres than other equipment options. This most likely should be considered when considering options for N management in the wheat crop.
Summary
With wheat sowing quickly approaching for many and field preparation nearing completion it is important to consider your nitrogen management options. Delayed N application allows for flexibility in management plan and depending on source utilized may increase application efficiency over pre-plant applications requiring a tillage implement. As fertilizer prices continue to remain high it is also important to consider the likely increase in N use efficiency due to applying N closer to when N requirement is peaking. Controlling cost while continuing to maximize output is imperative to sustainable profitability in crop production.
Any Question or Comments please feel free to reach out me.
Brian Arnall b.arnall@okstate.edu
Can Grain Sorghum Wait on Nitrogen?
Michaela Smith, Masters student under advisement of B. Arnall
Brian Arnall, Precision Nutrient Management Specialist
Grain sorghum producers in Oklahoma are challenged greatly by their environment and sporadic rainfall patterns, which diminish as the season progresses. These uncontrollable variables influence timing of nitrogen (N) application and nitrogen use efficiency. Using rainfall events as an incorporation method forces producers to apply before the event regardless of its intensity or delay application until field conditions are acceptable while anxiously waiting for another rainfall event. When deciding to delay N application it’s important to know the effects on physiological development and grain yield.

Trial structure and breakdown
This study was conducted over the 2020 growing season consisting four locations, including one double cropping system following wheat. Ten in-season applications were made using ammonium nitrate (AN) as the N source at a rate of 90 lbs. ac. Using AN as the N source reduced the risk of nitrogen loss through the process of volatilization as the goal of the research was to test the plant not the fertilizer. A pre-plant treatment served as the standard check, while in-season applications were initiated at 21 Days After Planting (DAP) and applications made sequentially at 7-day intervals. A non-fertilized check was included to the study to confirm locations were responsive to N fertilized applications Hybrid, plant date, and seeding rate can be found in Table 1.

Physiological Response to Application Timing
Two of the four locations demonstrated an effect to physiological development and maturity with the delay of nitrogen application. A delay in heading by a one to two-week period was observed at Perkins and Lahoma for applications made after May 21st (Table 2.). This delay in heading contributed to similar delay in maturity and potential harvest date. At Perkins decreased plant height was observed in the pre-plant plot and was associated with the onset of late season nitrogen deficiency (Figure 2). While this response was unexpected, the impact of nitrogen deficiency experienced early in the crop growth on the root and shoot growth has been well documented in many species. As a plant experiences nitrogen limitations growth changes from above ground to the below ground parts (roots) in an attempt to alleviate nitrogen stress. This increase in root growth could contribute to a more efficient uptake of nitrogen and decrease loss. In contrast to Figure 2, pre-plant application is shorter than compared to later season applications, this could be a result of inadequate N uptake thus leading to N loss by leaching, whereas later applications had increased root growth for efficient N interception and uptake.


Yield Response to Application Timing
Response of N was observed at all locations (Figure 3), while the delay of nitrogen varied in its effects across all locations. Grain yield from each N application was compared back to the pre-plant application to evaluate the effects of timing. All four locations responded positively to N fertilizer. At both LCB and Lahoma grain yield was maintained with applications made as late as 42 to 63 DAP respectively before any negative trend in grain yield was observed. Perkins was the only locations to have a statistically significant increase in grain yield due to delayed N applications. At this site, which is a sandy loam, waiting until 42 DAP resulted in a 15 bushel increase over the pre-plant plot. Now Alva which was double crop showed that rainfall is key. At this site, none of the in-season treatments made it up the level of the pre-plant. The reason for this will be discussed further below.

Influence of Rainfall
The loss in grain yield at Perkins in the pre-plant application could likely be reflective of nitrogen loss due to leaching. Pre-plant applications have been well documented in the aspect loss as a result of crop requirement and early physiological development. Long term mesonet rainfall data depicts a decline in the probability of rainfall with the progression of the growing season across all locations. In early season the probability of 0.5 inches of rainfall ranges from 8 to 10% respectively for LCB, Lahoma, and Perkins, and dramatically decline to percentages at low as 5% in mid-July during grain filling period. For Alva rainfall probability is substantially lower as its season was initiated during the drier months, which depicted a probability of 6% for 0.5 inches of rainfall, and 4.5% for 1 inch for early season rainfall crucial for pre-plant incorporation and crop establishment. These probabilities drop considerably compared to regular season as the months progress onward, mid to late August probability for 0.5 inches ranges from 0.8 to 11.5%, while for a 1 inch is 0 to 6.9%. Past weather data provided by the mesonet illustrates how later in the season rainfall and its amount is variable, suggesting that in a double crop scenario delayed application is not recommended while it is in regular season crop due to the increased chance of rainfall probability.
Summary
The purpose of this study was to evaluate the impacts of delayed nitrogen application in grain sorghum. In order to develop an accurate conclusion additional site years are required, although current data could suggest delaying nitrogen application for full season grain sorghum is possible without a detrimental loss in grain yield. This means producers have time to evaluate the crop and market to determine if more inputs are needed and economical, while allowing implementation of technologies such as the N-Rich Strip and SBNRC.
If you have any questions for comments please reach out.
Brian Arnall
b.arnall@okstate.edu
405.744.1722
Acknowledgement of EDC Ag Products Co LLC for support of this project.
Nitrogen timing in a winter wheat forage system
Bronc Finch Ph.D. student under the leadership of D.B. Arnall
Brian Arnall, Precision Nutrient Management Specialist.
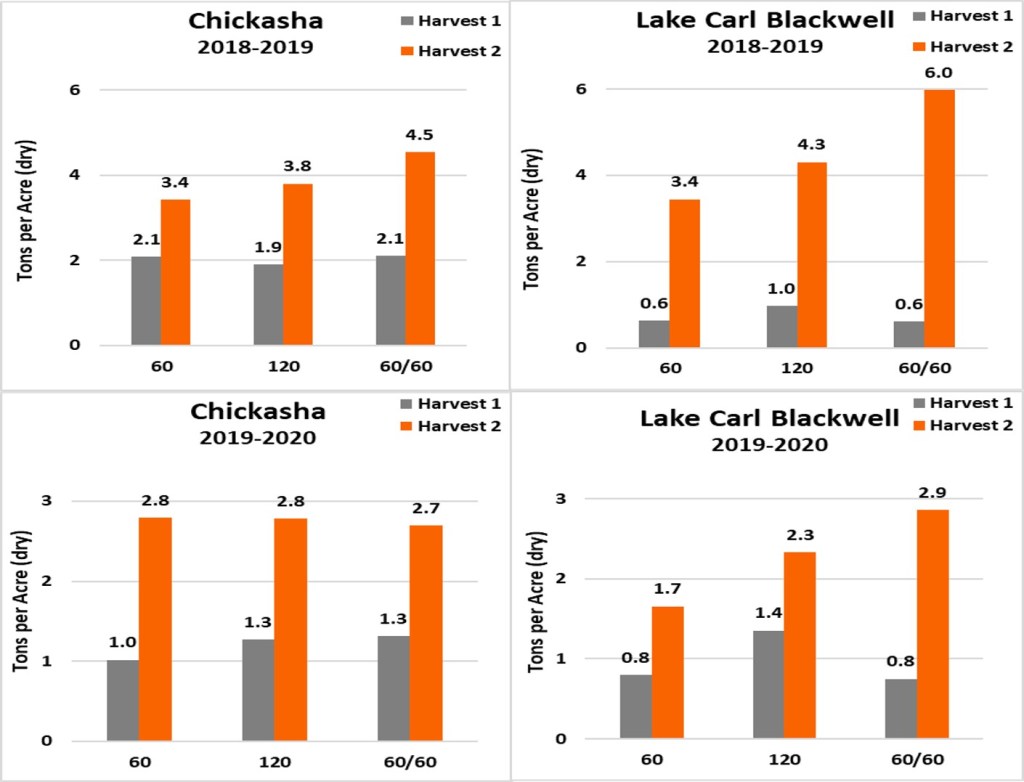
The recent weather conditions have caused a delay in the ability to top-dress winter wheat in some parts of Oklahoma. Despite this delay, conditions have still been good for growth, which means a steady increase towards the hollow stem and jointing stages. As these stages approach, or have passed, many concerns have been raised about the decision to apply nitrogen to increase spring forage production of winter wheat. A study conducted over the past two years at Oklahoma State University, in cooperation with Noble Research Institute has had the opportunity to evaluate how a nitrogen application at or just after hollow stem impacts the forage production of winter wheat. This study was set up with three fertilizer treatments of a 60 lb N pre-plant only, 120 lb N pre-plant only, and a 60 lb pre-plant and 60 lb top-dress applications. In the first season of the trial, 2018-2019, the fertilizer application was applied shortly after the wheat achieved the Feekes 6 stage (hollow-stem) due to rain and other conditions preventing a timely top-dress. In the 2019-2020 season of the trial the treatments were applied at a more ideal time, near the end of February and beginning of March before hollow stem. For this study the first cutting was targeted for just prior to hollow stem and the second cutting conducted at early boot stage.
In 2018-2019 the additional 60 lb N applied in the 120 lb N pre-plant increased the dry biomass production in the second harvest by 0.4 and 0.9 tons per acre above the 60 lbs treatment at Chickasha and Lake Carl Blackwell, respectively (Figure 1). The delay of the additional 60 lbs of N increased the yield by an additional 0.7 and 1.7 tons per acre, respectively. The 2019-2020 season showed similar results at the Lake Carl Blackwell location in the second harvest, where the additional 60 lbs N at pre-plant increased biomass yield by 0.6 tons, with the delaying of the additional 60 lbs increasing biomass yield by 0.6 more as compared to the same rate when applied at pre-plant. But that additional yield gained with the split application came at cost as the 120 pre-plant resulted in 0.6 tons more in the first harvest. These results suggest that more N was needed in the pre-plant and top-dress application. The 2019-2020 Chickasha trial showed little difference in rate, more than likely 60 lbs N was enough maximize forage yield.
Total biomass production for 2018-19 winter wheat forage (Figure 2) showed to have a greater increase in total biomass production when the N was split applied with the second application being made shortly after hollow stem. The split application increased total biomass production by as much as 1.3 tons per acre more than the same rate applied as all pre-plant. The 2019-2020 year total biomass production shows to be about the same whether the N was applied all pre or split and applied in February.

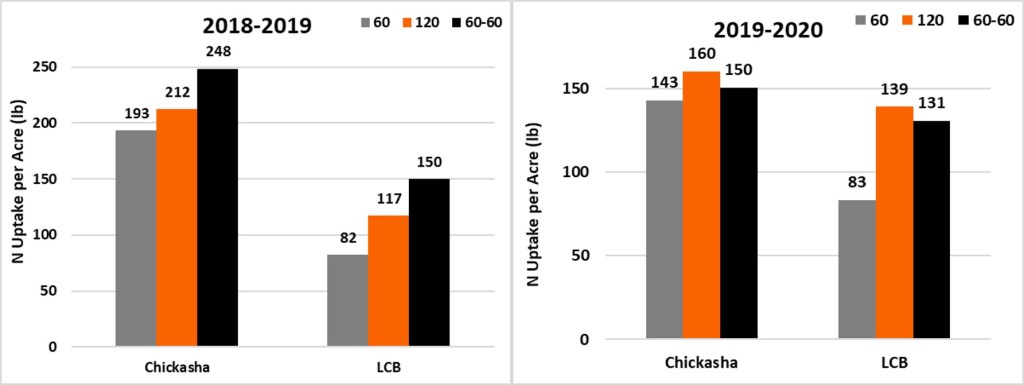
Figure 3 documents N uptake of winter wheat biomass for both years continues the same trend as total biomass. Nitrogen uptake can be directly related to protein as the calculation for protein is %N * 6.25. In all cases uptake was greater than applied. In 2018-2019 split application increased over all nitrogen uptake. Much like the yield of 2019-2020 the N uptake was not significantly impacted by the timing of the N application.
Although the assessment of N application made at or after hollow stem in a winter wheat forage system was not an objective of this study, the circumstances have given a unique opportunity to evaluate the outcome. In the 2018-2019 trials when top-dress application was applied at or just following the hollow stem, yield and nitrogen uptake were both increased over the equivalent pre-plant application. For the 2019-2020 season where N was applied at the planned time yields and N uptake were equivalent to the pre-plant. While this data is not conclusive it does indicate the producers can apply N fertilizer to winter wheat forage at or after hollow stem and successfully increase both forage yield and nitrogen uptake.
For questions or comments please feel free to reach out.
Brian Arnall
b.arnall@okstate.edu
405.744.1722
Acknowledgement of LSB Industries for support of these projects.
Value of in-season application for grain only wheat production.
Data used in this blog is summarized from work by
Joao Souza, under the leadership of D.B. Arnall
Lawrence Aula, under the leadership of W.R. Raun
Key Points
- Wheat is highly resilient and can endure nitrogen stress for a significant period of time and fully recover.
- Delaying all nitrogen until the Feekes 5 to Feekes 7 time frame resulted in improved yields over the pre-plant 32% of the time and a loss of yield 5%. However, grain protein was improved 82% of the time with delayed nitrogen.
- It is better to delay nitrogen application to avoid conditions conducive to N loss.
Historically winter wheat producers have utilized pre-plant nitrogen (N) fertilizer application due to efficiency of time and the lower cost of the primary N source, anhydrous ammonia. However, as the growing cycle of winter wheat is approximately 9 months long with only 80% of the total N accumulation reached by flowering. Research as shown that N applied prior to planting is more likely to be lost due to leaching or denitrification. Researchers at Oklahoma State University have invested significant efforts in evaluating N management strategies. This blog will present the data from multiple trials which allowed for the comparison of nitrogen applied pre-plant versus in-season. The trials were conducted over a four-year period at multiple locations across central Oklahoma.
Delayed Nitrogen – NH4NO3
This study was started in the fall of 2016 and concluded with the 2020 wheat harvest. In all, twelve trials were established and achieved maturity. This study was designed to evaluate the recovery of winter wheat grain yield and protein after the crop was N stressed. Treatments included an untreated check, pre-plant application and ten in-season treatments. The application of in-season treatments was initiated when N deficiency was confirmed and treatments were applied in progressive order every seven growing days to the point of 63 growing days after visual deficiency (DAVD). A growing degree days is any day that the average daily temperature is at or above 40⁰ F. Ammonium nitrate (NH4NO3) was applied at a rate of 90 lbs N ac-1 for all treatments.
Nitrogen response was observed at eleven of the twelve locations, and those sites will be the focus of this review. Nitrogen applications were started ranging from Nov. 10th to Mar. 7th for 0 DAVD and, concluded with 63 DAVD occurring between mid-February and early-May. The analysis of the data evaluated the yield and protein of the in-season applications compared to both the pre-plant application and the application made at the first sign of N deficiency, 0DAVD.
Across the eleven responsive years, the pre-plant application never outperformed the 0DAVD in terms of grain yield or protein. In fact, across all location if the in-season application was made prior to the end of March, the yield and protein was equal to or better than pre-plant applications. Four out of elevens sites, yield was significantly improved with in-season applications, and protein was improved in ten out of eleven locations. For the ten site/years that had applications in March, the mid-March application of 90 lbs of N, which is about the stage of hollow stem (Feekes 6), statistically increased yield four times and protein nine times compared to the pre-plant treatment.
The studies objective was to evaluate how long the crop could be deficient and fully recover. There was no relation between when the crop became deficient and when the crop could no longer recover. Yield as maintained as long as the N was applied by late March, or just before the flag leaf is visible (Feekes 8), grain yield was the same as if applied on the first day of deficiency. However, if the N was delayed to March protein was increased six out of the eleven locations.

Delayed Nitrogen – Urea
A mirror study to the Delayed Nitrogen – NH4NO3 was established in the fall of 2018 and concluded with the 2020 harvest. This study was placed next to the NH4NO3 and treatments applied on the same days using the same rate (90 lbs N ac-1) applied as urea to evaluate efficiency of urea applications over a range of dates.
Three of the four locations produced a positive response to N fertilizer and documented similar results as the NH4NO3 project. Across these three sites in-season N was always equal to the pre-plant rate if applied before the flag leaf is visible. In addition, if the urea was applied just after hollow stem, not only was yield maintained but protein was significantly increased compared to both the pre-plant and 0DAVD treatments at all three responsive sites.
Split Rate Nitrogen – NH4NO3
This study looked at multiple rates and times of N application but for this factsheet we will focus on a small set of treatments. Performed over two years and four total sites this project looked at split application of N versus a one-time application, 45/45 split or 90 lbs of N. Application timing was 0, 30, 60, 90, 120 growing days (GDD>0), trying to have applications at planting in December, February, March and April. In three of the four sites the 90 day application produced the greatest yield and protein for both 45/45 and 90 treatments. In this study the one-time application of 90 lbs N ac-1 out yielded the 45/45 split in two of the four years and was equal the other two. The 90 day application of 90 lbs N ac-1 produced a higher protein concentration at all sites compared to the 45/45 split applied on the same date.
Nitrogen Rate by Time – Urea Source
This study evaluated four rates of N (0, 40, 80, 120 lbs N) applied at three times (30 days pre-plant, pre-plant, and Feekes 5) using urea. Feekes 5 is the growth stage prior to hollow stem when the wheats leaf sheaths are becoming strongly erect. This project was completed over two locations for two years, however of the four site/years only three statistically responded to N fertilizer. In those three responsive trials the Feekes 5 application grain yield was equal to pre-plant once, greater than pre-plant once, and less than pre-plant once. The grain protein was only statistically different between the pre-plant and Feekes 5 once, with an increase in protein with late N. The one location with yield loss can be likely attributed to N loss from urea volatilization. The urea was applied on no-till immediately after a heavy rainfall with no substantial precipitation occurring for a week after application.
Summary
This factsheet summarizes four separate research projects which can contribute data from 24 trials to evaluate the application of in-season N compared to pre-plant N, see Table 1. Of these 24 site/years we can draw conclusions from the 22 that responded to N fertilizer applications. Across these trials applying all N pre-plant resulted in the highest grain yield once, applying all N in-season near or after hollow stem resulted in an increase in grain yield above that of the pre-plant seven times. However, the delaying of N application until hollow stem resulted in a significant increase in grain protein concentration at 18 of the 22 trials.
These results are significant for the winter wheat growers of the southern Great Plains as this research documented not only the ability but the necessity to move away from pre-plant and fall N applications for winter wheat grain production. The window for N application is likely much greater than most wheat producers would have considered. This work showed that not only could N be delayed and yield not sacrificed but, when delayed; yield will be maintained and protein concentration increased.
The final conclusion is that the timing of N application should not be based upon the presence of N deficiency or calendar date. Rather the timing should be based upon the weather and enviroment during application. While many of the projects used NH4NO3 as the N source to limit the impact of N loss via volatilization, the primary source for in-season nitrogen in the region are dry urea and urea ammonium nitrate (UAN) solution. Both of these sources have well documented loss due volatilization. The location from the Nitrogen Rate by Time trial which Feekes 5 applications were statisically below the pre-plant application supports this. This data set provides signifiant evidence that the optimum application window is quite wide and allowing producers more flexiabltiy to avoid those environments which will likely lead to N losses.
Special thanks to EDC Ag Products Co LLC for suppling NH4NO3 used in the delayed N project.