Home » IPM
Category Archives: IPM
Monitoring for Cotton Jassid: A Potential New Threat to Oklahoma Cotton
Ashleigh M. Faris, Maxwell Smith, & Jenny Dudak
While the cotton jassid (Amrasca biguttula), also known as the Two-Spot Cotton Leafhopper, has not yet been detected in Oklahoma, its rapid expansion across the Southern Cotton Belt in 2025 makes it a potential threat to the 2026 season. This pest is considered one of the most serious threats to U.S. cotton, with the potential for significant yield losses in untreated fields. Stay informed on if the cotton jassid may be moving from the south and into Oklahoma by signing up for Texas A&M AgriLife Extension text alerts: text COTTON to 833.717.0325.
Identification: The “Two-Spot” Difference
The cotton jassid can be confused with the native potato leafhopper, but its unique markings are the key to early detection (Figure 1).
- Adults: Approximately 1/8 inch (2mm) long, wedge-shaped, and green.
- Key Markings: Look for two small black spots on the crown of the head and one black spot on the tip of each forewing. These spots on the head can sometimes fade but are generally visible under magnification; the spots on the wings will be present in adults and do not fade.
- Nymphs: Wingless and pale green. They are best known for their “crab-like” sideways movement when disturbed on the leaf surface. Any nymphs spotted should warrant a thorough scouting for adult cotton jassids and damage.

Figure 1. Cotton jassid adults (A) have one black dot on each wing and may have two small dots between their eyes (these dots on the crown can fade). The potato leafhopper (B), which is not a threat to cotton production but does occur in Oklahoma, does not have black dots on their wings or between their eyes. Image A courtesy of Isaac Esquivel, UF Extension, image B courtesy of DryBeanAgronomy.ca.
Biology and Host Range
The cotton jassid has a short life cycle, completing a generation in approximately two weeks under warm conditions.
- Reproduction: Eggs are inserted directly into leaf midveins and petioles, hatching in 3–4 days. Eggs will not be visible to the naked eye or through hand lens. Cotton jassids progress through 5 nymphal instars before becoming reproductive adults (Figure 2).
- Host Plants: This pest is polyphagous, meaning it feeds on many hosts. While cotton is a primary target, it also thrives on okra, eggplant, and ornamental hibiscus. It has also been found on native plants like Turk’s cap, as well as weeds like Ceasar weed and Florida pusley.
- 2025 Range on U.S. Cotton: The cotton jassid was detected on cotton in FL, GA, AL, MS, LA, TN, SC, NC, and TX. The TX detections in cotton were limited to southeastern TX in Grimes, Wharton, and Fort Bend counties. At the time of this article’s posting (March 2026), the cotton jassid has not been detected in OK.
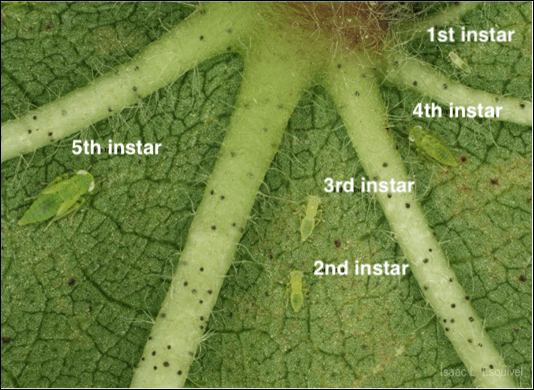
Figure 2. Cotton jassid nymphs on the underside of a cotton leaf. Image courtesy of Isaac Esquivel, UF Extension.
Damage: Recognizing Hopperburn
Unlike other leafhoppers, the cotton jassid injects a salivary toxin that disrupts the plant’s vascular system.
- Early Signs: Initial yellowing that resemble potassium deficiency with some upward curling of leaf margins (Figure 3, Rating 1).
- Progression: Characterized by hopperburn, a yellowing (chlorosis) that proceeds from leaf edges and turns red or brown as the tissue dies (Figure 3).
- Systemic Impact: Plants can go downhill quickly, often leading to complete desiccation and stunted growth (Figure 4).
A Hopperburn Injury Rating Scale has been developed by Extension Cotton Entomologists in the mid-South (Figure 3). You cannot let the cotton jassid get ahead of you. Once reddening starts on the leaf margins (Rating 2 in Figure 3) it is likely too late to rescue the cotton plant, damage will quickly progress and photosynthetic capabilities for the plant decline considerably.

Figure 3. Hopperburn injury rating scale for cotton jassid damage. Damage increases from none (0) to severe damage of desiccated leaf (5). Slight yellowing and upward curling of leaf is shown in Rating 1, with increased yellowing, cupping, and beginnings of reddened leaf margins in Rating 2. Insecticide action should be taken prior to reaching Rating 2. Ratings 3 – 5 show increased spread of reddening and desiccation. Images courtesy of Phillip Roberts (UGA Extension) and Scott Graham (AU Extension).

Figure 4. Cotton jassid hopperburn resulting in reddened, dried leaves (A) and stunted cotton plants (B). Image courtesy Isaac Esquivel, UF Extension.
Scouting Protocol
Scouting is mandatory for every cotton field in 2026 to prevent significant yield loss. Plants located at the edge of cotton fields can serve as good indicators, as cotton jassids will enter at field margins where damage is more likely to occur before further in field.
- Target Area: Inspect the undersides of leaves in the mid-to-upper canopy.
- Sample Leaf: Focus on the 4th mainstem leaf below the terminal, as this is where nymphs typically congregate.
- Visual Checks: Because adults fly quickly, count the flightless nymphs. Examine at least 25 leaves per field.
- Threshold: 1 cotton jassid per leaf, or early crop injury indicators (Figure 3, Rating 1) with cotton jassid confirmations nearby.
- Continue Scouting: Since green leaves are needed to fill bolls, growers should scout cotton up to at least 2 weeks prior to defoliation.
Management Guidance
Cultural Practices
- Plant Early: Trials indicate that earlier planting dates can help the crop “outrun” the peak pressure of migrating populations.
- Nutrient Management: Avoid excess Nitrogen, which attracts cotton jassids. Ensure adequate Potassium, as deficient plants crash much faster under cotton jassid stress.
- Varieties: Internationally, varieties with high trichome (hair) density on leaves offer natural resistance to feeding. However, varieties on the U.S. market are generally less hairy than those planted elsewhere. Currently, trials from 2025 do not indicate a varietal difference in terms of cotton jassid susceptibility.
Chemical Control
Based on 2025 research trials conducted by Mid-South Cotton Extension Entomologists, the following insecticides have shown varying levels of control (Table 1). Repeated insecticide applications may be warranted.
Table 1. Suggested foliar insecticides* and their observed control level for suppressing the cotton jassid. Efficacy lasted around 2 weeks.
| Control Level | Insecticides |
| High (>70% Control) | Carbine, Sefina, Sivanto, Bidrin, Venom, Plinazolin |
| Moderate (50-70%) | Transform, Centric, Assail, Orthene |
| Low (<50%) | Steward, Diamond, Bifenthrin, Admire Pro |
*Cotton jassids have shown resistance to every chemistry class in their native range; rotation of modes of action is critical. The mention, listing, or use of specific insecticides is not an endorsement of that product, nor is it a criticism of similar products not mentioned.
If you suspect cotton jassid activity or see hopperburn symptoms, contact the OSU Cotton IPM team: Maxwell Smith (maxwell.smith@okstate.edu), Ashleigh Faris (Ashleigh.faris@okstate.edu), and Jenny Dudak (jdudak@okstate.edu) immediately for confirmation. This team will be monitoring for the cotton jassid and will share updates on nearing threat, Oklahoma detections, and updated management guidance as it becomes available.
For more information on the cotton jassid in the U.S., click on this link to access Extension Factsheets, podcasts, and videos developed by Extension Entomologists managing the pest: https://drive.google.com/file/d/19IFT5c9b5JXEaBgf6X-weya07G56RXS5/view?usp=sharing.
Small Pest, Big Problems: Wheat Curl Mites and Wheat Streak Mosaic Virus Detected in Oklahoma
Ashleigh Faris, Cropping Systems Entomologist, IPM Coordinator
Meriem Aoun, Wheat Pathologist
Department of Entomology & Plant Pathology,
Oklahoma State University
Wheat Curl Mite (WCM) activity has been confirmed in Washita County, located in western Oklahoma. While the mites themselves are difficult to see, they can have a considerable impact on wheat health, primarily due to their role as vectors for several viral diseases such as wheat streak mosaic virus (WSMV). The Plant Disease and Insect Diagnostic Laboratory (PDIDL) has confirmed WSMV in the sample where WCM were detected in Washita County. This week, the PDIDL has also confirmed infection by WSMV in Blaine County (Canton, OK), McCurtain County (Garvin, OK), and Cleveland County (Noble, OK).
Identification
The Wheat Curl Mite is nearly invisible to the naked eye. At approximately 1/100 of an inch long, these pests require a 10x – 20x hand lens for proper identification.
- Appearance: They are white or cream-colored, cigar-shaped (cylindrical), and possess only four legs located near the head (Figure 1).
- Behavior: They are typically found in the protected areas of the plant, such as developing, youngest leaves or the furrows of the leaf surface. As the leaf unfurls, the mites migrate to the next emerging leaf.

Figure 1. Wheat curl mites and eggs on a wheat leaf (A, B), and mites on a maturing wheat kernel (C). Images courtesy G. Bauchan and R. Ochoa, USDA-ARS.
Biology and Life Cycle
Understanding the WCM life cycle is critical for preventative management:
- Rapid Reproduction: Under optimal temperatures (75° – 85°F), a WCM can complete its life cycle in 7 to 10 days. This allows populations to explode rapidly during warm autumns or springs.
- Dispersal: WCMs cannot fly; they rely entirely on wind currents to move from plant to plant or field to field. They crawl to the tips of leaves and hitchhike on the wind.
- Survival (The Green Bridge): WCMs are obligate parasites, meaning they require living green tissue to survive and reproduce. They persist through the summer on volunteer wheat and various perennial or annual grasses. This is known as the green bridge. If this bridge is not broken, mites move into the newly planted crop in the fall.
Damage and Virus Transmission
WCMs cause two types of damage:
- Direct Feeding: Mites suck sap from the leaf cells. This causes the edges of the leaf to roll inward (the curl part of WCM) (Figure 2). This curling provides a protected microclimate for the mites to reproduce. Heavy infestations can cause stunting and a slowed appearance in growth.
- Viral Vector (Primary Concern): The WCM is the sole vector for Wheat Streak Mosaic Virus (WSMV), High Plains Wheat Mosaic Virus (HPWMoV), and Triticum Mosaic Virus (TriMV).
- Symptoms: Infected plants show yellowing, mottled or streaked leaves, and severe stunting (Figures 3 & 4).
- Impact: If infection occurs in the fall, yield loss can be up to 100%. Spring infections are generally less damaging.
Scouting Techniques
Because the mites are so small, scouting focuses on leaf symptoms and having a hand lens:
- Check your Fields: Examine the youngest leaves of the wheat plant. Look for the characteristic inward rolling of the leaf edges (Figure 2).
- Use Magnification: Slowly unroll a suspect leaf and use a hand lens to look for tiny, white, slow-moving specks in the leaf furrows.
- Pattern of Infestation: Wind-dispersed mite infestations often start at the edge of a field (particularly edges adjacent to volunteer wheat or CRP land) and move inward in the direction of prevailing winds. Areas with infestations may show signs of yellowing and appear as patches distributed at random across the field (Figure 4).

Figure 2. Infestation of wheat curl mites on wheat results in tightly curled leaves and entrapment of subsequent leaves within the curl (A). After full leaf emergence, a tight curl at the leaf edge remains (B). Images courtesy of UNL Extension.

Figure 3. Wheat streak mosaic virus (WSMV) symptoms includeyellowing, mottled or streaked leaves. Image courtesy of Meriem Aoun, Oklahoma State University.

Figure 4. Plants at field margins, neighboring a wheat curl mite source, are the first to become infected with viruses of the Wheat Streak Mosaic Virus (WSMV) complex and develop symptoms, such as yellowing and streaking. Notice the gradient in color from the field edge (left) toward the center of the wheat field. Image courtesy of UNL Extension.
Management Recommendations
Currently, there are no effective rescue chemical treatments for WCM once symptoms appear in the field. Miticides generally do not reach the mites hidden inside the curled leaves. Management must be proactive:
- Manage volunteer wheat and grassy weeds: This is the most effective management tool to break the green bridge. Ensure all volunteer wheat and grassy weeds are completely dead (via tillage or herbicide) at least two weeks prior to planting the new crop. WCMs will starve within days without a living host.
- Delayed Planting: Planting wheat later in the fall reduces the window of time that mites must migrate into the crop and slows their reproduction rate as temperatures drop.
- Variety Selection: Some wheat varieties offer resistance or tolerance to WCM or WSMV. Consult the latest OSU variety trial data to select adapted varieties for north-central Oklahoma that carry these traits. Currently, Breakthrough is the most resistant OSU variety, which carries the WSMV resistance gene, Wsm1.
Brown Wheat Mite Activity in North Central Oklahoma
Ashleigh Faris, Cropping Systems Entomologist, IPM Coordinator
Department of Entomology & Plant Pathology,
Oklahoma State University
Following a period of dry weather, wheat growers in central Oklahoma are reporting activity of the Brown Wheat Mite (BWM). Unlike many other wheat pests, BWM thrives in drought conditions, and its damage can often be mistaken for moisture stress or nutrient deficiency.
Identification
The Brown Wheat Mite is small—about the size of a needle point—but is generally easier to spot than the Wheat Curl Mite because it is active on the leaf surface.
- Appearance: BWM has a dark red to brownish-black, oval-shaped body (Figure 1).
- Distinguishing Feature: Its front legs are significantly longer than its other three pairs of legs.
- Behavior: They are most active during the day, particularly in the afternoon, and will quickly drop to the ground if the plant is disturbed (Figure 2).

Figure 1. Brown wheat mite (BWM).

Figure 2. Brown wheat mites (BWM) on wheat. Image courtesy L. Galvin, OSU Extension.
Biology and Life Cycle
BWM populations consist entirely of females that produce offspring without mating (parthenogenesis), allowing for extremely rapid population growth under dry conditions. The BWM has a unique life cycle in that it can lay two types of eggs. Environmental conditions dictate when these two types of eggs are laid:
- Red Eggs: Laid during the growing season and hatch in about a week when conditions are favorable.
- White (Diapause) Eggs: Laid as temperatures rise and the crop matures. They are highly resistant and allow the population to survive the summer heat, hatching only when cooler, wetter weather arrives in the fall.
Damage
BWM damage is caused by the mites piercing plant cells and sucking out the plant nutrients.
- Symptoms: Initial damage appears as “stippling” (fine white or yellow spots) on the leaves. As feeding continues, leaves take on a silvery or bronzed appearance (Figure 3).
- Tipping: Heavy infestations cause the tips of the leaves to turn brown and die.
- Weather Interaction: Damage is most severe when plants are already under drought stress. Because both BWM damage and drought cause yellowing/browning, it is essential to confirm the presence of mites before treating.

Figure 3. Brown wheat mite (BWM) damage.
Scouting
Because BWM is highly mobile and drops when disturbed, careful scouting is required:
- Timing: Scout during the warmest part of the day when mites are most active on the upper leaves.
- The Paper Test: Gently but quickly shake or tap wheat plants over a white piece of paper or a white clipboard. Look for tiny dark specks moving across the surface.
- Economic Threshold: While thresholds vary based on crop value and moisture stress, research suggests a treatment threshold of 25 to 50 brown wheat mites per leaf in wheat that is 6 inches to 9 inches tall is economically warranted. An alternative estimation is “several hundred” per foot of row. If the wheat is severely stressed, the lower end of that threshold should be used.
Management Recommendations
- The “Rain” Factor: A significant, driving rain is often the most effective control for BWM. Rain can physically knock mites from the plant and promote fungal pathogens that naturally reduce the population.
- Chemical Control: If populations exceed the threshold and no rain is in the forecast, chemical intervention may be necessary. Know the cost of the treatment and value of your wheat so you can determine if an application is a worth return on investment.
- Effective Ingredients: Organophosphates (such as Dimethoate) have historically provided better control than many pyrethroids, as the latter can sometimes result in mite “flaring” or simply fail to provide adequate residual control.
- Coverage: High water volume is critical to ensure the insecticide reaches the mites, especially if they have moved toward the base of the plant.
- Pre-harvest Intervals & Grazing Restrictions: Always read and follow the label guidelines. For more on acaricides that can be applied in wheat see the Oklahoma State University Fact Sheet “Management of Insect and Mite Pests in Small Grains” (CR-7194).
- Cultural Practices: Since BWM thrives in dry, dusty conditions, maintaining good soil moisture and vigorous plant growth can help the crop tolerate feeding. Here’s to hoping for some rain soon in the forecast; we could really use it for lots of reasons in Oklahoma.
Check Your Wheat: Greenbugs Reported in Central Oklahoma
Ashleigh M. Faris
Cropping Systems Extension Entomologist
Department of Entomology & Plant Pathology
Oklahoma State University
Wheat producers in central Oklahoma are reporting the presence of the greenbug, Schizaphis graminum, in winter wheat fields. Greenbugs are one of the most important insect pests of wheat in the southern Great Plains and can occur from fall through spring. These aphids feed on plant sap and inject toxins into wheat plants, causing characteristic leaf discoloration and plant injury.
Early detection through field scouting is essential to determine whether populations are increasing and if an insecticide treatment is justified.
Greenbug Identification & Biology
Key identifying characteristics of greenbug (Figure 1):
- Small aphids (~1/16 inch long)
- Pale to lime-green body
- Dark green stripe down the middle of the back
- Dark tips on antennae and legs
- Found in colonies on the underside of wheat leaves

Greenbugs reproduce rapidly under favorable conditions (between 55° F and 95° F) and often occur in patches within fields rather than evenly distributed populations. During periods of cool weather, the greenbug may increase to enormous numbers, due to the absence of natural enemies, which develop significantly slower compared to greenbugs at such temperatures. On the other hand, cold weather can also influence aphid populations. However, this latest cold snap is not enough to eliminate greenbugs. It takes average temperatures below 20° F for at least a week to kill a substantial number of greenbugs in wheat.
Greenbug Damage in Wheat
Greenbugs damage wheat in two ways, through direct feeding and injection of toxic saliva. Greenbugs may also transmit barley yellow dwarf virus (BYDV), which can further reduce yield potential.
Typical early symptoms include small, reddish or copper spots on leaves (Figure 2) and yellowing around feeding sites. Advanced infestations will result in leaves turning yellow or orange, dead leaf tissue, stunted plants, and expanding patches of dead wheat. Heavy infestations may kill seedlings and reduce tillering, particularly during drought stress.

How to Scout for Greenbugs
The Glance-N-Go™ sampling system developed by Oklahoma State University can help determine whether aphid populations exceed economic thresholds. Download the Greenbug Glance N’ Go Sampler app for your smartphone. You will then input the control cost ($/Acre), crop value ($/Acre), and the Spring sampling window. Use a zig-zag or W-pattern (Figure 3) to scout your field, checking undersides of leaves at three tillers per stop for greenbugs and brown mummies. Use the app to record the numbers of these insects and sample until the app tells you to stop sampling or tells you treat. As temperatures warm, continue to scout regularly as greenbug populations may build.
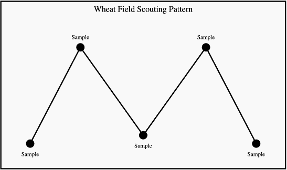
Scouting recommendations without the Greenbug Glance N’ Go Sampler app:
- Walk a W or zigzag pattern across the field.
- Examine 10–20 plants at each stop.
- Check:
- Underside of leaves
- Leaf midrib
- Base of tillers
- Record:
- Aphids per tiller
- Presence of aphid mummies (Figure 4)
- Beneficial insects
Beneficial Insects
Natural enemies frequently control aphid populations. While scouting for greenbug you should also look for lady beetles, lacewing larvae, hoverfly larvae, and parasitized aphids (“mummies”) (Figure 4). If beneficial insects are abundant, aphid populations may decline without insecticide treatment. Where there are one to two lady beetles (adults and larvae) per foot of row, or 15 to 20 percent of the greenbugs have been parasitized, control measures could be delayed until it is determined whether the greenbug population is continuing to increase.
Based on current wheat scouting, it appears that parasitoid numbers are low this 2026 season so continuing to scout for greenbug will be critical in responding to populations that go unchecked by beneficials.

Economic Threshold Guidelines
The simplest way to determine if action needs to be taken against greenbugs is to utilize the Glance-N-Go™ sampling system developed by Oklahoma State University. Approximate guidelines historically used in Oklahoma wheat can be found in Table 1 below.
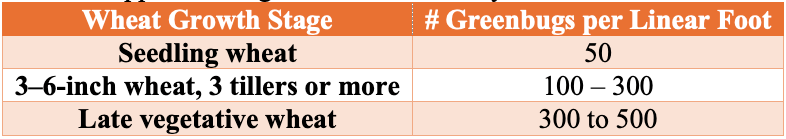
Thresholds are influenced by:
- Wheat growth stage
- Crop value
- Cost of treatment
- Presence of beneficial insects
Insecticides Labeled for Greenbugs in Wheat
Aphid feeding and insecticide performance are strongly influenced by temperature. Greenbugs tend to move higher on wheat plants during warm conditions but may move lower on the plant or below ground during cold weather, reducing exposure to insecticides. As a result, damaging populations are most often observed in late winter and early spring. Insecticides generally perform best when temperatures are above 50°F, and control may occur more slowly in cooler conditions (e.g., control at 45° F may take roughly twice as long as at 70° F). If applications must be made under cooler temperatures, use the highest labeled rate. Wheat grown under irrigation can typically tolerate higher greenbug populations than dryland wheat.
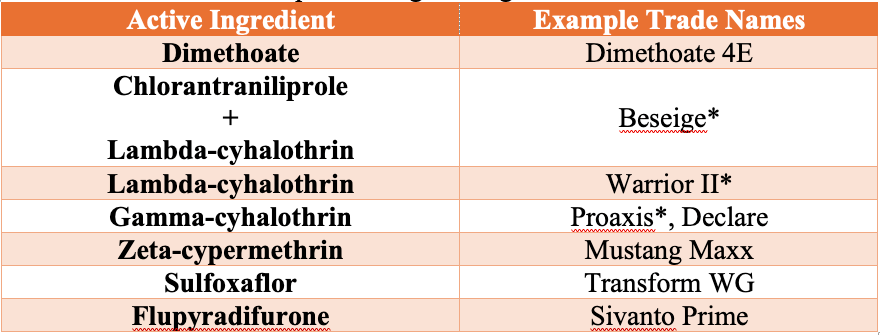
Always follow pesticide label directions, application sites, and rates. Be sure to read and follow the label for preharvest intervals (PHI) and restricted-entry intervals (REI). Use a minimum of 10 GPA by ground and 3 GPA by air (if labelled for aerial application) to ensure adequate coverage.
For assistance with aphid identification or treatment decisions, see OSU Fact Sheet EPP-7099 Small Grain Aphids in Oklahoma and Their Management, or contact your local OSU Extension office.
Cotton disease update: Reniform nematode – 08/25/2025
Maíra Duffeck, OSU Row Crops Extension Pathologist, Department of Entomology and Plant Pathology Oklahoma State University
Maxwell Smith, OSU IPM for Cotton Extension Specialist, Department of Entomology and Plant Pathology, Oklahoma State University
Jenny Dudak, OSU Extension Cotton Specialist, Department of Plant & Soil Sciences Oklahoma State University
Reniform nematode continues to be detected in cotton fields across Oklahoma. During the 2023 and 2024 growing seasons, a survey was conducted in 17 commercial cotton fields located in Tillman, Jackson, Grady, and Caddo counties to assess the presence of parasitic nematodes affecting cotton production. We collected soil samples in areas of the fields showing irregular and stunted cotton plants.
Out of the 17 soil samples collected, reniform nematode was detected in 5 fields, marking the first confirmed report of this pest in Oklahoma cotton. Notably, in one of the positive fields, the reniform nematode population reached 1,569 nematodes per 100 cm³ of soil; more than double the economic threshold of 700 nematodes per 100 cm³. In 2025, a soil sample from a cotton field in Jackson County already tested positive for the reniform nematode.
The reniform nematode, caused by Rotylenchulus reniformis, is one of the most important yield-limiting pathogens of cotton production in the southern U.S. In addition to cotton, the reniform nematode can reproduce on other field crops such as soybean, with yield loss estimates being greater in cotton than soybean. The reniform nematode is easy to introduce into new fields because of its unique ability to survive in a dehydrated state in dry soils. Therefore, it can be transported long distances on field equipment.
We suspect that parasitic nematodes, such as root-knot and reniform nematodes, are already present in many Oklahoma cotton fields, but the damage they cause often goes unnoticed. This is especially important for the reniform nematode, as yield losses can occur without obvious aboveground symptoms. For this reason, monitoring the distribution of this nematode across the cotton fields in Oklahoma is crucial to raise awareness of this emerging issue and to guide future management decisions.
Symptoms and Signs
The expression of symptoms depends on several factors, including the susceptibility of the cotton hybrid, nematode population levels, soil type, and for how long that field has been infested. Affected plants may show reduced growth, delayed flowering, fewer fruits, and smaller fruit size, which together contribute to yield losses in lint or pods. Unlike the southern root-knot nematode (Meloidogyne incognita), the reniform nematode does not induce gall formation on roots, making field diagnosis based solely on visible symptoms challenging. For this reason, soil testing through a nematode assay is often necessary for proper identification. In newly infested fields, stunted plants are typically the most noticeable sign (Figure 1).
Plan of Action
To address this issue, a statewide nematode survey is underway to document the presence, abundance, and geographic distribution of parasitic nematodes in Oklahoma cotton fields. The information generated from this survey will provide a foundation for developing and implementing economically viable strategies to manage this issue and protect cotton production in the state.
How to participate?
Oklahoma cotton growers interested in having their fields tested for parasitic nematodes have several ways to participate in this study:
- Schedule a field visit: Contact Dr. Maira Duffeck to arrange soil sample collection. She can be reached by phone at 347-205-2180 or by email at mairodr@okstate.edu.
- Drop off samples at the Peanut & Cotton Field Day: September 18, 2025, from 5:00–8:00 p.m. at the Caddo Research Station (28054 County Street 2540, Ft. Cobb)
- Drop off samples at the Cotton Field Day: September 25, 2025, from 8:30 a.m. to 1:00 p.m. at the Southwest Research & Extension Center (16721 US Hwy 283, Altus)
More information about dropping off samples on OSU field days can be found on the flyers shown in Figures 2 and 3. Growers can submit soil samples for analysis at no cost, as expenses are covered through a project funded by the Oklahoma Cotton Council in partnership with Cotton Incorporated.
How to collect soil samples for analysis?
- Soil samples should be collected from the root zone of the plants
- Collect 15–20 soil cores (6–8 inches deep) from across the field
- Growers should focus on areas of the field where plants are showing poor growth and development
- Mix the cores thoroughly, then place the mixed soil into a resealable plastic bag
- We need about 2 pints (1 kg) of soil for analysis
- Keep samples cool — store them in a refrigerator until the field day.
- If you collect soil samples from different fields, please label and add field information to the plastic bag accordingly



For Additional Information contact.
Dr. Maíra Duffeck mairodr@okstate.edu